Arylcyclohexylamine
Class of chemical compounds
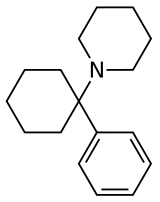
Arylcyclohexylamines , also known as arylcyclohexamines or arylcyclohexanamines , are a chemical class of pharmaceutical , designer , and experimental drugs .
History
Phencyclidine (PCP) is believed to be the first arylcyclohexylamine with recognized anesthetic properties, but several arylcyclohexylamines were described before PCP in the scientific literature, beginning with PCA (1-phenylcyclohexan-1-amine) the synthesis of which was first published in 1907. PCE was reported in 1953 and PCMo (4-(1-phenyl-cyclohexyl)-morpholine [1] see chart below for figure) in 1954, with PCMo described as a potent sedative. [2] Arylcyclohexylamine anesthetics were intensively investigated at Parke-Davis , beginning with the 1956 synthesis of phencyclidine and later the related compound ketamine . [2] The 1970s saw the debut of these compounds, especially PCP and its analogues , as illicitly used recreational drugs due to their dissociative hallucinogenic and euphoriant effects. Since that time, the class has been expanded by scientific research into stimulant , analgesic , and neuroprotective agents, and also by clandestine chemists in search of novel recreational drugs . [3] [4] [5]
Structure

An arylcyclohexylamine is composed of a cyclohexylamine unit with an aryl moiety attachment. The aryl group is positioned geminal to the amine . In the simplest cases, the aryl moiety is typically a phenyl ring , sometimes with additional substitution. The amine is usually not primary ; secondary amines such as methylamine or ethylamine, or tertiary cycloalkylamines such as piperidine and pyrrolidine , are the most commonly encountered N -substituents.
Pharmacology
Arylcyclohexylamines varyingly possess NMDA receptor antagonistic , [6] [7] dopamine reuptake inhibitory , [8] and μ-opioid receptor agonistic [9] properties. Additionally, σ receptor agonistic, [10] nACh receptor antagonistic, [11] and D 2 receptor agonistic [12] actions have been reported for some of these agents. Antagonism of the NMDA receptor confers anesthetic, anticonvulsant, neuroprotective, and dissociative effects; blockade of the dopamine transporter mediates stimulant and euphoriant effects as well as psychosis in high amounts; and activation of the μ-opioid receptor causes analgesic and euphoriant effects. Stimulation of the σ and D 2 receptors may also contribute to hallucinogenic and psychotomimetic effects. [12]
These are versatile agents with a wide range of possible pharmacological activities depending on the extent and range to which chemical modifications are implemented. [13] [14] [15] [16] [17] [18] [19] [20] [21] The various choice of substitutions that are made allows for "fine-tuning" of the pharmacological profile that results. As examples, BTCP is a selective dopamine reuptake inhibitor , [8] PCP is primarily an NMDA antagonist, [6] and BDPC is a potent μ-opioid agonist, [22] while PRE-084 is a selective sigma receptor agonist. [23] Thus, radically different pharmacology is possible through different structural combinations.
Notes on numbering
PCP itself is composed of three six-membered rings, which can each be substituted by a variety of groups. These are traditionally numbered in the older research as first the cyclohexyl ring, then the phenyl , and finally the piperidine ring, with the different rings represented by prime notation (') next to the number. For instance, 4-methyl-PCP, 4'-methyl-PCP and 4''-methyl-PCP are all known compounds, with similar activity but quite different potencies.

However, since the widespread sale of these compounds as grey-market designer drugs, nearly all such compounds that have come to prominence either have a bare cyclohexyl ring or a 2-ketocyclohexyl ring, while the piperidine is replaced by a variety of alkyl or cycloalkyl amines and most substitution has taken place on the phenyl ring. Consequently it is common for widely used phenyl substituted analogues such as 3'-MeO-PCP and 3'-MeO-PCE to be referred to as 3-MeO-PCP and 3-MeO-PCE without the prime, even though this is technically incorrect and could lead to confusion.
List of arylcyclohexylamines
| Structures | Compound | Aryl Substituent | N Group | Cyclohexyl ring | CAS number |
|---|---|---|---|---|---|

|
PCA [24] | Phenyl | NH 2 | - | 1934-71-0 |
|
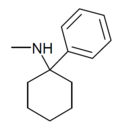
PCM [24] | Phenyl | Methylamino | - | 2201-16-3 |

|
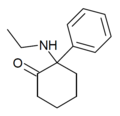
Eticyclidine | Phenyl | Ethylamino | - | 2201-15-2 |

|
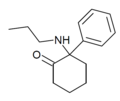
PCPr [25] | Phenyl | n -Propylamino | - | 18949-81-0 |
|
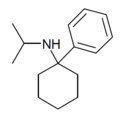
PCiP | Phenyl | Isopropylamino | - | 1195-42-2 |

|
PCAL [26] | Phenyl | Allylamino | - | 2185-95-7 |
|
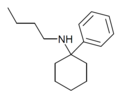
PCBu | Phenyl | n -Butylamino | - | 73166-29-7 |

|
PCEOH | Phenyl | Hydroxyethylamino | - | 2201-22-1 |

|
PCMEA [27] | Phenyl | Methoxyethylamino | - | 2201-57-2 |
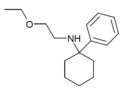
|
PCEEA | Phenyl | Ethoxyethylamino | - | 1072895-05-6 |
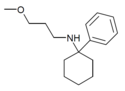
|
PCMPA | Phenyl | Methoxypropylamino | - | 2201-58-3 |

|
PCDM [24] | Phenyl | Dimethylamino | - | 2201-17-4 |

|
Dieticyclidine | Phenyl | Diethylamino | - | 2201-19-6 |

|
2-HO-PCP [6] | Phenyl | Piperidine | 2-Hydroxy | 94852-58-1 |
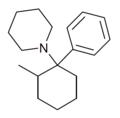
|
2-Me-PCP [28] | Phenyl | Piperidine | 2-Methyl | 59397-29-4 |

|
2-MeO-PCP [29] | Phenyl | Piperidine | 2-Methoxy | 78636-34-7 |
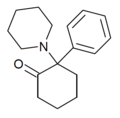
|
2-Keto-PCP | Phenyl | Piperidine | 2-Keto | 101688-16-8 |
|
Eticyclidone ("O-PCE") | Phenyl | Ethylamino | 2-Keto | 6740-82-5 |
|
2-Keto-PCPr | Phenyl | n -Propylamino | 2-Keto | |

|
4-Methyl-PCP | Phenyl | Piperidine | 4-Methyl | 19420-52-1 |

|
4-Keto-PCP [30] | Phenyl | Piperidine | 4-Keto | 65620-13-5 |
|
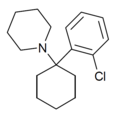
2'-Cl-PCP | o -Chlorophenyl | Piperidine | - | 2201-31-2 |

|
3'-Cl-PCP | m -Chlorophenyl | Piperidine | - | 2201-32-3 |
|
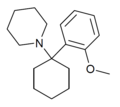
2'-MeO-PCP | o -Methoxyphenyl | Piperidine | - | 2201-34-5 |

|
3'-F-PCP [31] | m -Fluorophenyl | Piperidine | - | 89156-99-0 |
|
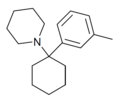
3'-Me-PCP [32] | m -Tolyl | Piperidine | - | 2201-30-1 |

|
3'-Me-PCPy | m -Tolyl | Pyrrolidine | - | 1622348-63-3 |

|
3'-NH 2 -PCP | m -Aminophenyl | Piperidine | - | 72242-00-3 |
|
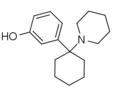
3'-HO-PCP | m -Hydroxyphenyl | Piperidine | - | 79787-43-2 |
|
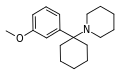
3'-MeO-PCP | m -Methoxyphenyl | Piperidine | - | 72242-03-6 |

|
3',4'-MD-PCP | 3,4-Methylenedioxyphenyl | Piperidine | - | |
|
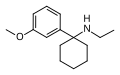
3'-MeO-PCE | m -Methoxyphenyl | Ethylamino | - | 1364933-80-1 |
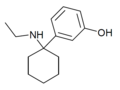
|
3'-HO-PCE | m -Hydroxyphenyl | Ethylamino | - | |
|
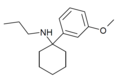
3'-MeO-PCPr | m -Methoxyphenyl | n -Propylamino | - | 1364933-81-2 |

|
3'-HO-PCPr | m -Hydroxyphenyl | n -Propylamino | - | |
|
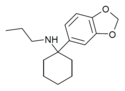
3',4'-MD-PCPr | 3,4-Methylenedioxyphenyl | n -Propylamino | - | |
|
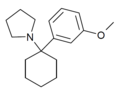
3'-MeO-PCPy [32] | m -Methoxyphenyl | Pyrrolidine | - | 1364933-79-8 |

|
4'-HO-PCP | p -Hydroxyphenyl | Piperidine | - | 66568-88-5 |

|
Methoxydine (4'-MeO-PCP) | p -Methoxyphenyl | Piperidine | - | 2201-35-6 |

|
4'-MeO-PCE | p -Methoxyphenyl | Ethylamino | - | |

|
4'-F-PCP [31] | p -Fluorophenyl | Piperidine | - | 22904-99-0 |
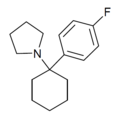
|
4'-F-PCPy | p -Fluorophenyl | Pyrrolidine | - | |
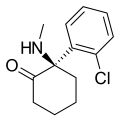
|
Arketamine | o -Chlorophenyl | Methylamino | 2-Keto | 33643-49-1 |

|
Deschloroketamine | Phenyl | Methylamino | 2-Keto | 7063-30-1 |

|
Esketamine | o -Chlorophenyl | Methylamino | 2-Keto | 33643-46-8 |

|
Ketamine | o -Chlorophenyl | Methylamino | 2-Keto | 6740-88-1 |

|
Hydroxynorketamine | o -Chlorophenyl | NH 2 | 2-Keto, 6-Hydroxy | 81395-70-2 |

|
Ethketamine | o -Chlorophenyl | Ethylamino | 2-Keto | 1354634-10-8 |

|
NPNK | o -Chlorophenyl | n -Propylamino | 2-Keto | 2749326-65-4 |

|
Methoxyketamine | o -Methoxyphenyl | Methylamino | 2-Keto | 7063-51-6 |
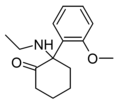
|
2-MeO-NEK [33] | o -Methoxyphenyl | Ethylamino | 2-Keto | |
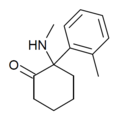
|
oMDCK [34] | o -Tolyl | Methylamino | 2-Keto | 7063-37-8 |
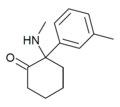
|
mMDCK | m -Tolyl | Methylamino | 2-Keto | |

|
meta -Ketamine | m -Chlorophenyl | Methylamino | 2-Keto | 7063-53-8 |

|
iso -Ketamine | o -Chlorophenyl | Methylamino | 4-Keto | |
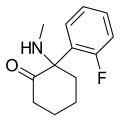
|
2-Fluorodeschloroketamine | o -Fluorophenyl | Methylamino | 2-Keto | 111982-50-4 |

|
3-Fluorodeschloroketamine | m -Fluorophenyl | Methylamino | 2-Keto | 2657761-23-2 |

|
Bromoketamine | o -Bromophenyl | Methylamino | 2-Keto | 120807-70-7 |

|
TFMDCK | o -Trifluoromethylphenyl | Methylamino | 2-Keto | 1782149-73-8 |

|
SN 35210 [35] | o -Chlorophenyl | Carbomethoxybutylamino | 2-Keto | 1450615-41-4 |
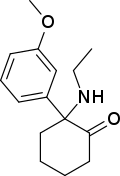
|
Methoxetamine | m -Methoxyphenyl | Ethylamino | 2-Keto | 1239943-76-0 |

|
Methoxmetamine | m -Methoxyphenyl | Methylamino | 2-Keto | 1781829-56-8 |
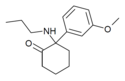
|
Methoxpropamine | m -Methoxyphenyl | n -Propylamino | 2-Keto | 2504100-71-2 |
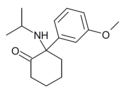
|
MXiPr | m -Methoxyphenyl | i -Propylamino | 2-Keto | |

|
Ethoxetamine | m -Ethoxyphenyl | Ethylamino | 2-Keto | |

|
Deoxymethoxetamine (3-Me-2'-Oxo-PCE) | m -Tolyl | Ethylamino | 2-Keto | 2666932-45-0 |

|
Br-MXE | 2-bromo-5-methoxyphenyl | Ethylamino | 2-Keto | |
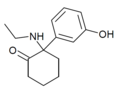
|
Hydroxetamine (HXE) | m -Hydroxyphenyl | Ethylamino | 2-Keto | 1620054-73-0 |
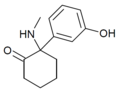
|
HXM | m -Hydroxyphenyl | Methylamino | 2-Keto | |
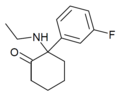
|
Fluorexetamine (FXE) | m -Fluorophenyl | Ethylamino | 2-Keto | |
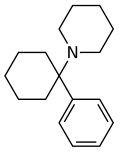
|
Phencyclidine (PCP) | Phenyl | Piperidine | - | 77-10-1 |
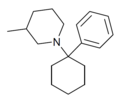
|
PC3MP | Phenyl | 3-Methylpiperidine | - | 2201-41-4 |

|
PC4MP | Phenyl | 4-Methylpiperidine | - | 2201-42-5 |

|
Rolicyclidine (PCPy) | Phenyl | Pyrrolidine | - | 2201-39-0 |
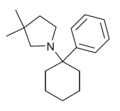
|
PCDMPy | Phenyl | 3,3-Dimethylpyrrolidine | - | |
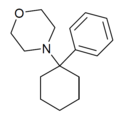
|
PCMo | Phenyl | Morpholine | - | 2201-40-3 |

|
Methoxy-PCM [7] (2'-MeO-PCMo) | o -Methoxyphenyl | Morpholine | - | 1314323-88-0 |

|
3'-MeO-PCMo | m -Methoxyphenyl | Morpholine | - | 138873-80-0 |

|
4'-MeO-PCMo | p -Methoxyphenyl | Morpholine | - | |

|
Methyl-PCM [36] (4'-Me-PCMo) | p -Tolyl | Morpholine | - | 120803-52-3 |

|
Hydroxy-methyl-PCM | 2-Methyl-4-hydroxyphenyl | Morpholine | - | 1314323-89-1 |
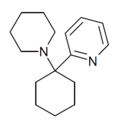
|
PYCP [37] | 2-Pyridinyl | Piperidine | - | |
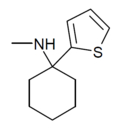
|
TCM | 2-Thienyl | Methylamino | - | 139401-07-3 |
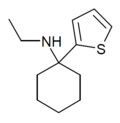
|
TCE | 2-Thienyl | Ethylamino | - | 101589-62-2 |
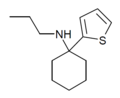
|
TCPr [38] | 2-Thienyl | Propylamino | - | |

|
Tenocyclidine (TCP) | 2-Thienyl | Piperidine | - | 21500-98-1 |

|
T3CP | 3-Thienyl | Piperidine | - | 19420-50-9 |
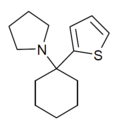
|
TCPy | 2-Thienyl | Pyrrolidine | - | 22912-13-6 |

|
Tiletamine | 2-Thienyl | Ethylamino | 2-Keto | 14176-49-9 |
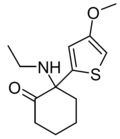
|
MXTE | 4-Methoxy-2-thienyl | Ethylamino | 2-Keto | |

|
Gacyclidine | 2-Thienyl | Piperidine | 2-Methyl | 68134-81-6 |
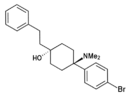
|
BDPC | p -Bromophenyl | Dimethylamino | 4-Phenethyl-4-hydroxy | 77239-98-6 |

|
C-8813 | p -Bromophenyl | Dimethylamino | 4-(thiophen-2-yl)ethyl-4-hydroxy | 616898-54-5 |

|
Dimetamine [39] | p -Tolyl | Dimethylamino | 4-Keto | 65619-06-9 |

|
3''-OH-2'-Me-PCP [40] | o -Tolyl | 3-Hydroxypiperidine | - | |

|
4''-Ph-4''-OH-PCP [41] | Phenyl | 4-Phenyl-4-hydroxypiperidine | - | 77179-39-6 |

|
BTCP [42] | Benzothiophen-2-yl | Piperidine | - | 112726-66-6 |

|
BTCPy [10] | Benzothiophen-2-yl | Pyrrolidine | - | |
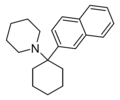
|
GK-189 [43] | Naphthalen-2-yl | Piperidine | - | 81490-58-6 |
Related compounds
Other similar compounds exist where the base ring has been varied, or the amine chain replaced with other groups. [44] More cycloalkane ring sizes have been experimented with than just purely thinking in terms of the cyclohexylamine. The cyclopentyl homologue of PCP is active with around one-tenth the potency, [45] while the cycloheptyl and cyclooctyl derivatives are inactive, though some substituted arylcycloheptylamines retain activity. [46] The requisite cycloalkylketone is reacted with PhMgBr; 3° alcohol is then reacted with NaN 3 ; azide then reduced with LAH. Then in the final step the piperidine ring is constructed with 1-5-dibromo-pentane. [47] Other compounds are known where the cyclohexyl base ring is replaced by rings such as norbornyl, adamantyl, [48] tetralin, oxane, thiane [49] or piperidine. [50] Conformationally constrained analogs have been prepared and researched by Morieti et al. [51]
| Structure | Compound | Aryl Substituent | N Group | Base ring | CAS number |
|---|---|---|---|---|---|

|
PCPEP | Phenyl | Piperidine | Cyclopentyl | 23036-19-3 |

|
3F-PCHEPy | 3-Fluorophenyl | Pyrrolidine | Cycloheptyl | |

|
3-MeO-PBCHP | 3-Methoxyphenyl | Piperidine | Bicyclo[2.2.1]heptane | |

|
PADP (P2AP) | Phenyl | Piperidine | Adamantyl | 72241-99-7 |
|
3-MeO-PTP | 3-Methoxyphenyl | Piperidine | Tetralin | |

|
HHFA | Fused phenyl | Amino | Hexahydrofluorene | |

|
DHPQ | Phenyl | Decahydroquinoline | ||

|
POXP | Phenyl | Piperidine | Oxane | |

|
PTHP | Phenyl | Piperidine | Thiane | |

|
MPBPip | Phenyl | Piperidine | N-Methylpiperidine | 36882-04-9 |

|
BnCP | Benzyl | Piperidine | Cyclohexyl | 22912-07-8 |

|
YNCP | Ethynyl | Piperidine | Cyclohexyl | 51165-02-7 |

|
ALCP | Allyl | Piperidine | Cyclohexyl | 7418-80-6 |
|
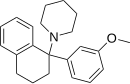
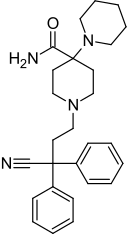
Piritramide | Replaced by carboxamide | Piperidine | N-(3-cyano-3,3-diphenylpropyl)piperidine | 302-41-0 |
|
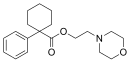
PRE-084 | Phenyl | Morpholinylethylcarboxylate | Cyclohexyl | 138847-85-5 |

|
Clofenciclan | p-Chlorophenyl | Diethylaminoethoxy | Cyclohexyl | 5632-52-0 |
References
- ↑ "4-(1-phenyl-cyclohexyl)-morpholine" . CAS Number Search - chemsrc.com . chemsrc . Retrieved 15 March 2021 .
- 1 2 Morris H, Wallach J (2014). "From PCP to MXE: a comprehensive review of the non-medical use of dissociative drugs". Drug Testing and Analysis . 6 (7–8): 614–32. doi : 10.1002/dta.1620 . PMID 24678061 .
- ↑ Valter K, Arrizabalaga P (1998). Designer Drugs Directory . Elsevier. ISBN 0-444-20525-X .
- ↑ Wallach J, Brandt SD (August 2018). "Phencyclidine-Based New Psychoactive Substances". New Psychoactive Substances . Handbook of Experimental Pharmacology. Vol. 252. pp. 261–303. doi : 10.1007/164_2018_124 . ISBN 978-3-030-10560-0 . PMID 30105474 .
- ↑ Wallach J, Brandt SD (2018). "1,2-Diarylethylamine- and Ketamine-Based New Psychoactive Substances". New Psychoactive Substances . Handbook of Experimental Pharmacology. Vol. 252. pp. 305–352. doi : 10.1007/164_2018_148 . ISBN 978-3-030-10560-0 . PMID 30196446 .
- 1 2 3 Ahmadi A, Mahmoudi A (2005). "Synthesis and biological properties of 2-hydroxy-1-(1-phenyltetralyl)piperidine and some of its intermediates as derivatives of phencyclidine". Arzneimittel-Forschung . 55 (9): 528–32. doi : 10.1055/s-0031-1296900 . PMID 16229117 . S2CID 29006399 .
- 1 2 Ahmadi A, Khalili M, Hajikhani R, Naserbakht M (April 2011). "New morpholine analogues of phencyclidine: chemical synthesis and pain perception in rats". Pharmacology, Biochemistry, and Behavior . 98 (2): 227–33. doi : 10.1016/j.pbb.2010.12.019 . PMID 21215770 . S2CID 24650035 .
- 1 2 Chaudieu I, Vignon J, Chicheportiche M, Kamenka JM, Trouiller G, Chicheportiche R (March 1989). "Role of the aromatic group in the inhibition of phencyclidine binding and dopamine uptake by PCP analogs". Pharmacology, Biochemistry, and Behavior . 32 (3): 699–705. doi : 10.1016/0091-3057(89)90020-8 . PMID 2544905 . S2CID 7672918 .
- ↑ Itzhak Y, Simon EJ (August 1984). "A novel phencyclidine analog interacts selectively with mu opioid receptors". The Journal of Pharmacology and Experimental Therapeutics . 230 (2): 383–6. PMID 6086884 .
- 1 2 He XS, Raymon LP, Mattson MV, Eldefrawi ME, de Costa BR (April 1993). "Synthesis and biological evaluation of 1-[1-(2-benzo[b]thienyl)cyclohexyl]piperidine homologues at dopamine-uptake and phencyclidine- and sigma-binding sites". Journal of Medicinal Chemistry . 36 (9): 1188–93. doi : 10.1021/jm00061a009 . PMID 8098066 .
- ↑ Eterović VA, Lu R, Eakin AE, Rodríguez AD, Ferchmin PA (December 1999). "Determinants of phencyclidine potency on the nicotinic acetylcholine receptors from muscle and electric organ" . Cellular and Molecular Neurobiology . 19 (6): 745–57. doi : 10.1023/A:1006905106834 . PMID 10456235 . S2CID 22266452 .
- 1 2 Seeman P, Ko F, Tallerico T (September 2005). "Dopamine receptor contribution to the action of PCP, LSD and ketamine psychotomimetics" . Molecular Psychiatry . 10 (9): 877–83. doi : 10.1038/sj.mp.4001682 . PMID 15852061 .
- ↑ al-Deeb OA (May 1996). "New analgesics derived from the phencyclidine analogue thienylcyclidine". Arzneimittel-Forschung . 46 (5): 505–8. PMID 8737636 .
- ↑ Ahmadi A, Khalili M, Hajikhani R, Hosseini H, Afshin N, Nahri-Niknafs B (March 2012). "Synthesis and study the analgesic effects of new analogues of ketamine on female wistar rats". Medicinal Chemistry . 8 (2): 246–51. doi : 10.2174/157340612800493683 . PMID 22385170 . S2CID 42842315 .
- ↑ Ahmadi A, Khalili M, Abbassi S, Javadi M, Mahmoudi A, Hajikhani R (2009). "Synthesis and study on analgesic effects of 1-[1-(4-methylphenyl) (cyclohexyl)] 4-piperidinol and 1-[1-(4-methoxyphenyl) (cyclohexyl)] 4-piperidinol as two new phencyclidine derivatives". Arzneimittel-Forschung . 59 (4): 202–6. doi : 10.1055/s-0031-1296386 . PMID 19517897 . S2CID 5402425 .
- ↑ Ahmadi A, Khalili M, Marami S, Ghadiri A, Nahri-Niknafs B (January 2014). "Synthesis and pain perception of new analogues of phencyclidine in NMRI male mice". Mini Reviews in Medicinal Chemistry . 14 (1): 64–71. doi : 10.2174/1389557513666131119203551 . PMID 24251803 .
- ↑ Ahmadi A, Solati J, Hajikhani R, Pakzad S (2011). "Synthesis and analgesic effects of new pyrrole derivatives of phencyclidine in mice". Arzneimittel-Forschung . 61 (5): 296–300. doi : 10.1055/s-0031-1296202 . PMID 21755813 . S2CID 24287727 .
- ↑ Ahmadi A, Khalili M, Hajikhani R, Barghi L, Mihandoust F (2010). "Synthesis and determination of chronic and acute thermal and chemical pain activities of a new derivative of phencyclidine in rats" . Iranian Journal of Pharmaceutical Research . 9 (4): 379–85. PMC 3870061 . PMID 24381602 .
- ↑ Ahmadi A, Khalili M, Mihandoust F, Barghi L (2010). "Synthesis and determination of acute and chronic pain activities of 1-[1-(3-methylphenyl) (tetralyl)]piperidine as a new derivative of phencyclidine via tail immersion and formalin tests". Arzneimittel-Forschung . 60 (1): 30–5. doi : 10.1055/s-0031-1296245 . PMID 20184224 . S2CID 23966936 .
- ↑ Hajikhani R, Ahmadi A, Naderi N, Yaghoobi K, Shirazizand Z, Rezaee NM, Niknafs BN (2012). "Effect of phencyclidine derivatives on anxiety-like behavior using an elevated-plus maze test in mice". Advances in Clinical and Experimental Medicine . 21 (3): 307–12. PMID 23214193 .
- ↑ Ahmadi A, Khalili M, Mirza B, Mohammadi-Diz M, Azami-Lorestani F, Ghaderi P, Nahri-Niknafs B (2017). "Synthesis and Antinociception Activities of Some Novel Derivatives of Phencyclidine with Substituted Aminobenzothiazoles". Mini Reviews in Medicinal Chemistry . 17 (1): 78–84. doi : 10.2174/1389557516666160428112532 . PMID 27121715 .
- ↑ Lednicer D, VonVoigtlander PF (October 1979). "4-(p-Bromophenyl)-4-(dimethylamino)-1-phenethylcyclohexanol, an extremely potent respresentative of a new analgesic series". Journal of Medicinal Chemistry . 22 (10): 1157–8. doi : 10.1021/jm00196a001 . PMID 513062 .
- ↑ Maurice T, Su TP, Parish DW, Nabeshima T, Privat A (December 1994). "PRE-084, a sigma selective PCP derivative, attenuates MK-801-induced impairment of learning in mice" . Pharmacology, Biochemistry, and Behavior . 49 (4): 859–69. doi : 10.1016/0091-3057(94)90235-6 . PMID 7886099 . S2CID 54306053 .
- 1 2 3 Thurkauf A, de Costa B, Yamaguchi S, Mattson MV, Jacobson AE, Rice KC, Rogawski MA (May 1990). "Synthesis and anticonvulsant activity of 1-phenylcyclohexylamine analogues". Journal of Medicinal Chemistry . 33 (5): 1452–8. doi : 10.1021/jm00167a027 . PMID 2329567 .
- ↑ Sauer C, Peters FT, Staack RF, Fritschi G, Maurer HH (April 2008). "Metabolism and toxicological detection of a new designer drug, N-(1-phenylcyclohexyl)propanamine, in rat urine using gas chromatography-mass spectrometry". Journal of Chromatography A . 1186 (1–2): 380–90. doi : 10.1016/j.chroma.2007.11.002 . PMID 18035363 .
- ↑ Kalir A, Teomy S, Amir A, Fuchs P, Lee SA, Holsztynska EJ, et al. (October 1984). "N-allyl analogues of phencyclidine: chemical synthesis and pharmacological properties". Journal of Medicinal Chemistry . 27 (10): 1267–71. doi : 10.1021/jm00376a006 . PMID 6481761 .
- ↑ Sauer C, Peters FT, Schwaninger AE, Meyer MR, Maurer HH (February 2009). "Investigations on the cytochrome P450 (CYP) isoenzymes involved in the metabolism of the designer drugs N-(1-phenyl cyclohexyl)-2-ethoxyethanamine and N-(1-phenylcyclohexyl)-2-methoxyethanamine". Biochemical Pharmacology . 77 (3): 444–50. doi : 10.1016/j.bcp.2008.10.024 . PMID 19022226 .
- ↑ Iorio MA, Tomassini L, Mattson MV, George C, Jacobson AE (August 1991). "Synthesis, stereochemistry, and biological activity of the 1-(1-phenyl-2-methylcyclohexyl)piperidines and the 1-(1-phenyl-4-methylcyclohexyl)piperidines. Absolute configuration of the potent trans-(-)-1-(1-phenyl-2-methylcyclohexyl)piperidine". Journal of Medicinal Chemistry . 34 (8): 2615–23. doi : 10.1021/jm00112a041 . PMID 1875352 .
- ↑ Ahmadi A, Mahmoudi A (2006). "Synthesis with improved yield and study on the analgesic effect of 2-methoxyphencyclidine". Arzneimittel-Forschung . 56 (5): 346–50. doi : 10.1055/s-0031-1296732 . PMID 16821645 . S2CID 10370245 .
- ↑ Ortiz DM, Custodio RJ, Abiero A, Botanas CJ, Sayson LV, Kim M, et al. (July 2021). "The dopaminergic alterations induced by 4-F-PCP and 4-Keto-PCP may enhance their drug-induced rewarding and reinforcing effects: Implications for abuse". Addiction Biology . 26 (4): e12981. doi : 10.1111/adb.12981 . PMID 33135332 . S2CID 226234538 .
- 1 2 Ogunbadeniyi AM, Adejare A (2002). "Syntheses of fluorinated phencyclidine analogs". Journal of Fluorine Chemistry . 114 : 39–42. doi : 10.1016/S0022-1139(01)00565-6 .
- 1 2 Wallach J, De Paoli G, Adejare A, Brandt SD (2013). "Preparation and analytical characterization of 1-(1-phenylcyclohexyl)piperidine (PCP) and 1-(1-phenylcyclohexyl)pyrrolidine (PCPy) analogues". Drug Testing and Analysis . 6 (7–8): 633–50. doi : 10.1002/dta.1468 . PMID 23554350 .
- ↑ Sayson LV, Botanas CJ, Custodio RJ, Abiero A, Kim M, Lee HJ, et al. (July 2019). "The novel methoxetamine analogs N-ethylnorketamine hydrochloride (NENK), 2-MeO-N-ethylketamine hydrochloride (2-MeO-NEK), and 4-MeO-N-ethylketamine hydrochloride (4-MeO-NEK) elicit rapid antidepressant effects via activation of AMPA and 5-HT2 receptors". Psychopharmacology . 236 (7): 2201–2210. doi : 10.1007/s00213-019-05219-x . PMID 30891619 . S2CID 83463722 .
- ↑ WO 2021134086 , Kruegel AC, Sames D, Hashimoto K, "Arylcyclohexylamine derivatives and their use in the treatment of psychiatric disorders", published 1 July 2021, assigned to Gilgamesh Pharmaceuticals, Inc. and The Trustees Of Columbia University In The City Of New York.
- ↑ Harvey M, Sleigh J, Voss L, Pruijn F, Jose J, Gamage S, Denny W (2015). "Determination of the Hypnotic Potency in Rats of the Novel Ketamine Ester Analogue SN 35210". Pharmacology . 96 (5–6): 226–32. doi : 10.1159/000439598 . PMID 26352278 . S2CID 36017002 .
- ↑ Ahmadi A, Khalili M, Hajikhani R, Naserbakht M (2011). "Synthesis and determination of acute and chronic pain activities of 1-[1-(4-methylphenyl) (cyclohexyl)] morpholine as a new phencyclidine derivative in rats". Arzneimittel-Forschung . 61 (2): 92–7. doi : 10.1055/s-0031-1296173 . PMID 21428243 . S2CID 8094521 .
- ↑ Zarantonello P, Bettini E, Paio A, Simoncelli C, Terreni S, Cardullo F (April 2011). "Novel analogues of ketamine and phencyclidine as NMDA receptor antagonists". Bioorganic & Medicinal Chemistry Letters . 21 (7): 2059–63. doi : 10.1016/j.bmcl.2011.02.009 . PMID 21334205 .
- ↑ Wallach J, Colestock T, Cicali B, Elliott SP, Kavanagh PV, Adejare A, et al. (August 2016). "Syntheses and analytical characterizations of N-alkyl-arylcyclohexylamines" (PDF) . Drug Testing and Analysis . 8 (8): 801–15. doi : 10.1002/dta.1861 . PMID 26360516 . S2CID 1599386 .
- ↑ Lednicer D, VonVoigtlander PF, Emmert DE (April 1980). "4-Amino-4-arylcyclohexanones and their derivatives, a novel class of analgesics. 1. Modification of the aryl ring". Journal of Medicinal Chemistry . 23 (4): 424–30. doi : 10.1021/jm00178a014 . PMID 7381841 .
- ↑ Ahmadi A, Solati J, Hajikhani R, Onagh M, Javadi M (2010). "Synthesis and analgesic effects of 1-[1-(2-methylphenyl)(cyclohexyl)]-3-piperidinol as a new derivative of phencyclidine in mice". Arzneimittel-Forschung . 60 (8): 492–6. doi : 10.1055/s-0031-1296317 . PMID 20863005 . S2CID 24803623 .
- ↑ Itzhak Y, Kalir A, Weissman BA, Cohen S (May 1981). "New analgesic drugs derived from phencyclidine". Journal of Medicinal Chemistry . 24 (5): 496–9. doi : 10.1021/jm00137a004 . PMID 7241506 .
- ↑ Vignon J, Pinet V, Cerruti C, Kamenka JM, Chicheportiche R (April 1988). "[3H]N-[1-(2-benzo(b)thiophenyl)cyclohexyl]piperidine ([3H]BTCP): a new phencyclidine analog selective for the dopamine uptake complex". European Journal of Pharmacology . 148 (3): 427–36. doi : 10.1016/0014-2999(88)90122-7 . PMID 3384005 .
- ↑ Kamenka JM, et al. Substituted cyclic amines and pharmaceutical composition containing them. Patent US5248686, 28 September 1993
- ↑ Wallach JV. Structure activity relationship (SAR) studies of arylcycloalkylamines as N-methyl-D-aspartate receptor antagonists. PhD. Thesis, University of the Sciences in Philadelphia, 19 Dec 2014.
- ↑ Shulgin AT, Mac Lean DE (1976). "Illicit synthesis of phencyclidine (PCP) and several of its analogs". Clinical Toxicology . 9 (4): 553–60. doi : 10.3109/15563657608988157 . PMID 975751 .
- ↑ Sun S, Wallach J, Adejare A (2014). "Syntheses and N-methyl-D-aspartate receptor antagonist pharmacology of fluorinated arylcycloheptylamines". Medicinal Chemistry . Shariqah (United Arab Emirates). 10 (8): 843–52. doi : 10.2174/1573406410666140428104444 . PMID 24773376 .
- ↑ McQuinn RL, Cone EJ, Shannon HE, Su TP (December 1981). "Structure-activity relationships of the cycloalkyl ring of phencyclidine". Journal of Medicinal Chemistry . 24 (12): 1429–32. doi : 10.1021/jm00144a011 . PMID 7310819 .
- ↑ Eaton TA, Houk KN, Watkins SF, Fronczek FR (April 1983). "Geometries and conformational processes in phencyclidine and a rigid adamantyl analogue: variable-temperature NMR, X-ray crystallographic, and molecular mechanics studies". Journal of Medicinal Chemistry . 26 (4): 479–86. doi : 10.1021/jm00358a005 . PMID 6834381 .
- ↑ Sisco E, Urbas A. Identification and Characterization of Designer Phencyclidines (PCPs) in Forensic Casework
- ↑ Gerhard O, Eberhard E. 4-amino-piperidines. US3311624A
- ↑ Moriarty RM, Enache LA, Zhao L, Gilardi R, Mattson MV, Prakash O (February 1998). "Rigid phencyclidine analogues. Binding to the phencyclidine and sigma 1 receptors". Journal of Medicinal Chemistry . 41 (4): 468–77. doi : 10.1021/jm970059p . PMID 9484497 .
Further reading
- Morris H, Wallach J (2014). "From PCP to MXE: a comprehensive review of the non-medical use of dissociative drugs". Drug Testing and Analysis . 6 (7–8): 614–32. doi : 10.1002/dta.1620 . PMID 24678061 .
External links
- Synthesis and Effects of PCP Analogs
- Interview with a Ketamine Chemist
- New Drugs: Designing Novel Arylcyclohexylamines
| Inhalational | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Injection |
|
||||||||||||||
|
|
Psychedelics
( 5-HT 2A agonists) |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Dissociatives
( NMDAR antagonists ) |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Deliriants
( mAChR antagonists ) |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Others |
|
| Adamantanes | |
|---|---|
| Adenosine antagonists | |
| Alkylamines | |
| Ampakines | |
| Arylcyclohexylamines | |
| Benzazepines | |
| Cathinones |
|
| Cholinergics |
|
| Convulsants | |
| Eugeroics | |
| Oxazolines | |
| Phenethylamines |
|
| Phenylmorpholines | |
| Piperazines | |
| Piperidines |
|
| Pyrrolidines | |
| Racetams | |
| Tropanes |
|
| Tryptamines | |
| Others |
|
| AMPAR |
|
|---|---|
| KAR |
|
| NMDAR |
|
|
DAT
( DRIs ) |
|
|||||||
|---|---|---|---|---|---|---|---|---|
|
NET
( NRIs ) |
|
|||||||
|
SERT
( SRIs ) |
|
|||||||
| VMATs | ||||||||
| Others |
|
|||||||