Seproxetine
Chemical compound
|
|
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | 4–16 days |
| Identifiers | |
|
|
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard ( EPA ) | |
| Chemical and physical data | |
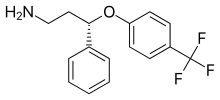
| Formula | C 16 H 16 F 3 N O |
| Molar mass | 295.305 g·mol −1 |
|
This article
needs additional citations for
verification
.
Please help
improve this article
by
adding citations to reliable sources
. Unsourced material may be challenged and removed.
Find sources: "Seproxetine" – news · newspapers · books · scholar · JSTOR ( July 2019 ) ( Learn how and when to remove this template message ) |
Seproxetine , also known as ( S )-norfluoxetine , is a selective serotonin reuptake inhibitor (SSRI). [1] It is the S enantiomer of norfluoxetine, the main active metabolite of the widely used antidepressant fluoxetine ; but little is known about its pharmacological actions. Seproxetine was being investigated by Eli Lilly and Company as an antidepressant; however, cardiac side effects were discovered [2] and development was discontinued. [1]
References
- 1 2 "Seproxetine" . DrugBank . University of Alberta . Retrieved 10 August 2016 .
- ↑ "Seproxetine" . Inxight Drugs . National Center for Advancing Translational Sciences (NCATS) . Retrieved 20 April 2023 .
|
Anxiolytics
(
N05B
)
|
|
|---|---|
| 5-HT 1A R agonists | |
| GABA A R PAMs |
|
|
Gabapentinoids
( α 2 δ VDCC blockers ) |
|
| Antidepressants |
|
|
Sympatholytics
( Antiadrenergics ) |
|
| Others | |
|
|
DAT
( DRIs ) |
|
|||||||
|---|---|---|---|---|---|---|---|---|
|
NET
( NRIs ) |
|
|||||||
|
SERT
( SRIs ) |
|
|||||||
| VMATs | ||||||||
| Others |
|
|||||||
| Corporate directors | |
|---|---|
| Products |
|

|
This drug article relating to the nervous system is a stub . You can help Wikipedia by expanding it . |
