Tyrosine
Amino acid
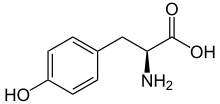
Skeletal formula
of
L
-tyrosine
|
||

L
-Tyrosine at physiological pH
|
||
|
||
| Names | ||
|---|---|---|
|
IUPAC name
(
S
)-Tyrosine
|
||
|
Other names
L
-2-Amino-3-(4-hydroxyphenyl)propanoic acid
|
||
| Identifiers | ||
|
||
|
3D model (
JSmol
)
|
||
| ChEBI | ||
| ChEMBL | ||
| ChemSpider |
|
|
| DrugBank |
|
|
| ECHA InfoCard |
100.000.419
|
|
| KEGG | ||
|
PubChem
CID
|
||
| UNII | ||
|
CompTox Dashboard
(
EPA
)
|
||
|
||
|
||
| Properties | ||
| C 9 H 11 N O 3 | ||
| Molar mass | 181.191 g·mol −1 | |
| .0453 g/100 mL | ||
|
Magnetic susceptibility
(
χ
)
|
-105.3·10 −6 cm 3 /mol | |
| Hazards | ||
| NFPA 704 (fire diamond) | ||
| Supplementary data page | ||
| Tyrosine (data page) | ||
|
Except where otherwise noted, data are given for materials in their
standard state
(at 25
°C [77
°F], 100
kPa).
|
L -Tyrosine or tyrosine (symbol Tyr or Y ) [2] or 4-hydroxyphenylalanine is one of the 20 standard amino acids that are used by cells to synthesize proteins . It is a non-essential amino acid with a polar side group . The word "tyrosine" is from the Greek tyrós , meaning cheese , as it was first discovered in 1846 by German chemist Justus von Liebig in the protein casein from cheese. [3] [4] It is called tyrosyl when referred to as a functional group or side chain. While tyrosine is generally classified as a hydrophobic amino acid, it is more hydrophilic than phenylalanine . [5] It is encoded by the codons UAC and UAU in messenger RNA .
Functions
Aside from being a proteinogenic amino acid , tyrosine has a special role by virtue of the phenol functionality. It occurs in proteins that are part of signal transduction processes and functions as a receiver of phosphate groups that are transferred by way of protein kinases . Phosphorylation of the hydroxyl group can change the activity of the target protein, or may form part of a signaling cascade via SH2 domain binding.
A tyrosine residue also plays an important role in photosynthesis . In chloroplasts ( photosystem II ), it acts as an electron donor in the reduction of oxidized chlorophyll . In this process, it loses the hydrogen atom of its phenolic OH-group. This radical is subsequently reduced in the photosystem II by the four core manganese clusters .
Dietary requirements and sources
The Dietary Reference Intake for tyrosine is usually estimated together with phenylalanine . It varies depending on an estimate method, however the ideal proportion of these two amino acids is considered to be 60:40 (phenylalanine:tyrosine) as a human body has such composition. [6] Tyrosine, which can also be synthesized in the body from phenylalanine, is found in many high- protein food products such as chicken , turkey , fish , milk , yogurt , cottage cheese , cheese , peanuts , almonds , pumpkin seeds , sesame seeds , soy protein and lima beans . [7] For example, the white of an egg has about 250 mg per egg, [8] while lean beef, lamb, pork, salmon, chicken, and turkey contain about 1 g per 3 ounces (85 g) portion. [8]
Biosynthesis

In plants and most microorganisms, tyrosine is produced via prephenate , an intermediate on the shikimate pathway . Prephenate is oxidatively decarboxylated with retention of the hydroxyl group to give p -hydroxyphenylpyruvate, which is transaminated using glutamate as the nitrogen source to give tyrosine and α-ketoglutarate .
Mammals synthesize tyrosine from the essential amino acid phenylalanine (Phe), which is derived from food. The conversion of Phe to Tyr is catalyzed by the enzyme phenylalanine hydroxylase , a monooxygenase. This enzyme catalyzes the reaction causing the addition of a hydroxyl group to the end of the 6-carbon aromatic ring of phenylalanine , such that it becomes tyrosine.
Metabolism

Phosphorylation and sulfation
Some of the tyrosine residues can be tagged (at the hydroxyl group) with a phosphate group ( phosphorylated ) by protein kinases . In its phosphorylated form, tyrosine is called phosphotyrosine . Tyrosine phosphorylation is considered to be one of the key steps in signal transduction and regulation of enzymatic activity. Phosphotyrosine can be detected through specific antibodies . Tyrosine residues may also be modified by the addition of a sulfate group, a process known as tyrosine sulfation . [9] Tyrosine sulfation is catalyzed by tyrosylprotein sulfotransferase (TPST). Like the phosphotyrosine antibodies mentioned above, antibodies have recently been described that specifically detect sulfotyrosine. [10]
Precursor to neurotransmitters and hormones
In dopaminergic cells in the brain , tyrosine is converted to L-DOPA by the enzyme tyrosine hydroxylase (TH). TH is the rate-limiting enzyme involved in the synthesis of the neurotransmitter dopamine . Dopamine can then be converted into other catecholamines , such as norepinephrine (noradrenaline) and epinephrine (adrenaline).
The thyroid hormones triiodothyronine (T 3 ) and thyroxine (T 4 ) in the colloid of the thyroid are also derived from tyrosine.
|
|
Precursor to other compounds
The latex of Papaver somniferum , the opium poppy, has been shown to convert tyrosine into the alkaloid morphine and the bio-synthetic pathway has been established from tyrosine to morphine by using Carbon-14 radio-labelled tyrosine to trace the in-vivo synthetic route. [14] Tyrosine ammonia lyase (TAL) is an enzyme in the natural phenols biosynthesis pathway. It transforms L-tyrosine into p-coumaric acid. Tyrosine is also the precursor to the pigment melanin . Tyrosine (or its precursor phenylalanine) is needed to synthesize the benzoquinone structure which forms part of coenzyme Q10 .
Degradation

The decomposition of L-tyrosine (syn. para -hydroxyphenylalanine) begins with an α-ketoglutarate dependent transamination through the tyrosine transaminase to para -hydroxyphenylpyruvate . The positional description para , abbreviated p , mean that the hydroxyl group and side chain on the phenyl ring are across from each other (see the illustration below).
The next oxidation step catalyzes by p -hydroxyphenylpyruvate dioxygenase and splitting off CO 2 homogentisate (2,5-dihydroxyphenyl-1-acetate). [15] In order to split the aromatic ring of homogentisate, a further dioxygenase, homogentisate 1,2-dioxygenase is required. Thereby, through the incorporation of a further O 2 molecule, maleylacetoacetate is created.
Fumarylacetoacetate is created by maleylacetoacetate cis - trans -isomerase through rotation of the carboxyl group created from the hydroxyl group via oxidation. This cis-trans -isomerase contains glutathione as a coenzyme . Fumarylacetoacetate is finally split by the enzyme fumarylacetoacetate hydrolase through the addition of a water molecule.
Thereby fumarate (also a metabolite of the citric acid cycle) and acetoacetate (3-ketobutyroate) are liberated. Acetoacetate is a ketone body , which is activated with succinyl-CoA, and thereafter it can be converted into acetyl-CoA , which in turn can be oxidized by the citric acid cycle or be used for fatty acid synthesis .
Phloretic acid is also a urinary metabolite of tyrosine in rats. [16]
Ortho- and meta-tyrosine

Three structural isomers of L-tyrosine are known. In addition to the common amino acid L-tyrosine, which is the para isomer ( para -tyr, p -tyr or 4-hydroxyphenylalanine), there are two additional regioisomers, namely meta -tyrosine (also known as 3-hydroxyphenylalanine , L- m -tyrosine , and m -tyr) and ortho -tyrosine ( o -tyr or 2-hydroxyphenylalanine), that occur in nature. The m -tyr and o -tyr isomers, which are rare, arise through non-enzymatic free-radical hydroxylation of phenylalanine under conditions of oxidative stress . [17] [18]
m-Tyrosine and analogues (rare in nature but available synthetically) have shown application in Parkinson's disease , Alzheimer's disease and arthritis . [19]
Medical use
Tyrosine is a precursor to neurotransmitters and increases plasma neurotransmitter levels (particularly dopamine and norepinephrine), [20] but has little if any effect on mood in normal subjects. [21] [22] [23] A number of studies have found tyrosine is useful during stress, cold, fatigue (in mice), [24] [25] prolonged work and sleep deprivation, [26] [27] with reductions in stress hormone levels, [28] reductions in stress-induced weight loss seen in animal trials, [25] and improvements in cognitive and physical performance [22] [29] [30] seen in human trials.
Tyrosine does not seem to have any significant effect on cognitive or physical performance in normal circumstances, [31] [32] but does help sustain working memory better during multitasking. [33] A 2015 systematic review found that "tyrosine loading acutely counteracts decrements in working memory and information processing that are induced by demanding situational conditions such as extreme weather or cognitive load " and therefore "tyrosine may benefit healthy individuals exposed to demanding situational conditions". [34]
Industrial synthesis
L-tyrosine and its derivatives ( L-DOPA , melanin , phenylpropanoids , and others) are used in pharmaceuticals , dietary supplements , and food additives . Two methods were formerly used to manufacture L-tyrosine. The first involves the extraction of the desired amino acid from protein hydrolysates using a chemical approach. The second utilizes enzymatic synthesis from phenolics, pyruvate, and ammonia through the use of tyrosine phenol-lyase . [35] Advances in genetic engineering and the advent of industrial fermentation have shifted the synthesis of L-tyrosine to the use of engineered strains of E. coli . [36] [35]
See also
References
- 1 2 Frey MN, Koetzle TF, Lehmann MS, Hamilton WC (1973). "Precision neutron diffraction structure determination of protein and nucleic acid components. X. A comparison between the crystal and molecular structures of L‐tyrosine and L‐tyrosine hydrochloride". J. Chem. Phys. 58 (6): 2547–2556. Bibcode : 1973JChPh..58.2547F . doi : 10.1063/1.1679537 .
- ↑ "Nomenclature and Symbolism for Amino Acids and Peptides" . IUPAC-IUB Joint Commission on Biochemical Nomenclature. 1983. Archived from the original on 9 October 2008 . Retrieved 5 March 2018 .
- ↑ "Tyrosine" . The Columbia Electronic Encyclopedia, 6th ed . Infoplease.com — Columbia University Press. 2007 . Retrieved 2008-04-20 .
- ↑ Harper D (2001). "Tyrosine" . Online Etymology Dictionary . Retrieved 2008-04-20 .
- ↑ "Amino Acids - Tyrosine" . www.biology.arizona.edu . Retrieved 2018-01-31 .
- ↑ Pencharz PB, Hsu JW, Ball RO (June 2007). "Aromatic amino acid requirements in healthy human subjects" . The Journal of Nutrition . 137 (6 Suppl 1): 1576S–1578S, discussion 1597S-1598S. doi : 10.1093/jn/137.6.1576S . PMID 17513429 .
- ↑ "Tyrosine" . University of Maryland Medical Center . Archived from the original on 2013-06-04 . Retrieved 2011-03-17 .
- 1 2 Top 10 Foods Highest in Tyrosine
- ↑ Hoffhines AJ, Damoc E, Bridges KG, Leary JA, Moore KL (December 2006). "Detection and purification of tyrosine-sulfated proteins using a novel anti-sulfotyrosine monoclonal antibody" . The Journal of Biological Chemistry . 281 (49): 37877–87. doi : 10.1074/jbc.M609398200 . PMC 1764208 . PMID 17046811 .
- ↑ Kanan Y, Hamilton RA, Sherry DM, Al-Ubaidi MR (December 2012). "Focus on molecules: sulfotyrosine" . Experimental Eye Research . 105 : 85–6. doi : 10.1016/j.exer.2012.02.014 . PMC 3629733 . PMID 22406006 .
- ↑ Broadley KJ (March 2010). "The vascular effects of trace amines and amphetamines". Pharmacology & Therapeutics . 125 (3): 363–375. doi : 10.1016/j.pharmthera.2009.11.005 . PMID 19948186 .
- ↑ Lindemann L, Hoener MC (May 2005). "A renaissance in trace amines inspired by a novel GPCR family". Trends in Pharmacological Sciences . 26 (5): 274–281. doi : 10.1016/j.tips.2005.03.007 . PMID 15860375 .
- ↑ Wang X, Li J, Dong G, Yue J (February 2014). "The endogenous substrates of brain CYP2D". European Journal of Pharmacology . 724 : 211–218. doi : 10.1016/j.ejphar.2013.12.025 . PMID 24374199 .
- ↑ Battersby, A. R.; Binks, R.; Harper, B. J. T. (1962-01-01). "692. Alkaloid biosynthesis. Part II. The biosynthesis of morphine" . Journal of the Chemical Society : 3534–3544. doi : 10.1039/JR9620003534 . ISSN 0368-1769 .
- ↑ Zea-Rey AV, Cruz-Camino H, Vazquez-Cantu DL, Gutiérrez-García VM, Santos-Guzmán J, Cantú-Reyna C (27 November 2017). "The Incidence of Transient Neonatal Tyrosinemia Within a Mexican Population" . Journal of Inborn Errors of Metabolism and Screening . 5 : 232640981774423. doi : 10.1177/2326409817744230 .
- ↑ Booth AN, Masri MS, Robbins DJ, Emerson OH, Jones FT, DeEds F (1960). "Urinary phenolic acid metabolities of tyrosine" . Journal of Biological Chemistry . 235 (9): 2649–2652. doi : 10.1016/S0021-9258(19)76930-0 .
- ↑ Molnár GA, Wagner Z, Markó L, Kó Szegi T, Mohás M, Kocsis B, et al. (November 2005). "Urinary ortho-tyrosine excretion in diabetes mellitus and renal failure: evidence for hydroxyl radical production" . Kidney International . 68 (5): 2281–7. doi : 10.1111/j.1523-1755.2005.00687.x . PMID 16221230 .
- ↑ Molnár GA, Nemes V, Biró Z, Ludány A, Wagner Z, Wittmann I (December 2005). "Accumulation of the hydroxyl free radical markers meta-, ortho-tyrosine and DOPA in cataractous lenses is accompanied by a lower protein and phenylalanine content of the water-soluble phase". Free Radical Research . 39 (12): 1359–66. doi : 10.1080/10715760500307107 . PMID 16298866 . S2CID 31154432 .
- ↑ Humphrey CE, Furegati M, Laumen K, La Vecchia L, Leutert T, Müller-Hartwieg JC, Vögtle M (2007). "Optimized Synthesis of L-m-Tyrosine Suitable for Chemical Scale-Up". Organic Process Research & Development . 11 (6): 1069–1075. doi : 10.1021/op700093y .
- ↑ Rasmussen DD, Ishizuka B, Quigley ME, Yen SS (October 1983). "Effects of tyrosine and tryptophan ingestion on plasma catecholamine and 3,4-dihydroxyphenylacetic acid concentrations". The Journal of Clinical Endocrinology and Metabolism . 57 (4): 760–3. doi : 10.1210/jcem-57-4-760 . PMID 6885965 .
- ↑ Leathwood PD, Pollet P (1982). "Diet-induced mood changes in normal populations". Journal of Psychiatric Research . 17 (2): 147–54. doi : 10.1016/0022-3956(82)90016-4 . PMID 6764931 .
- 1 2 Deijen JB, Orlebeke JF (1994). "Effect of tyrosine on cognitive function and blood pressure under stress". Brain Research Bulletin . 33 (3): 319–23. doi : 10.1016/0361-9230(94)90200-3 . PMID 8293316 . S2CID 33823121 .
- ↑ Lieberman HR, Corkin S, Spring BJ, Wurtman RJ, Growdon JH (August 1985). "The effects of dietary neurotransmitter precursors on human behavior". The American Journal of Clinical Nutrition . 42 (2): 366–70. doi : 10.1093/ajcn/42.2.366 . PMID 4025206 .
- ↑ Yeghiayan, S. K.; Luo, S.; Shukitt-Hale, B.; Lieberman, H. R. (2001). "Tyrosine improves behavioral and neurochemical deficits caused by cold exposure" . Physiology & Behavior . 72 (3): 311–316. doi : 10.1016/s0031-9384(00)00398-x . PMID 11274672 . S2CID 41813026 .
- 1 2 Hao S, Avraham Y, Bonne O, Berry EM (February 2001). "Separation-induced body weight loss, impairment in alternation behavior, and autonomic tone: effects of tyrosine". Pharmacology, Biochemistry, and Behavior . 68 (2): 273–81. doi : 10.1016/S0091-3057(00)00448-2 . PMID 11267632 . S2CID 46405659 .
- ↑ Magill RA, Waters WF, Bray GA, Volaufova J , Smith SR, Lieberman HR, et al. (August 2003). "Effects of tyrosine, phentermine, caffeine D-amphetamine, and placebo on cognitive and motor performance deficits during sleep deprivation". Nutritional Neuroscience . 6 (4): 237–46. doi : 10.1080/1028415031000120552 . PMID 12887140 . S2CID 21300076 .
- ↑ Neri DF, Wiegmann D, Stanny RR, Shappell SA, McCardie A, McKay DL (April 1995). "The effects of tyrosine on cognitive performance during extended wakefulness". Aviation, Space, and Environmental Medicine . 66 (4): 313–9. PMID 7794222 .
- ↑ Reinstein DK, Lehnert H, Wurtman RJ (December 1985). "Dietary tyrosine suppresses the rise in plasma corticosterone following acute stress in rats". Life Sciences . 37 (23): 2157–63. doi : 10.1016/0024-3205(85)90566-1 . PMID 4068899 .
- ↑ Deijen JB, Wientjes CJ, Vullinghs HF, Cloin PA, Langefeld JJ (January 1999). "Tyrosine improves cognitive performance and reduces blood pressure in cadets after one week of a combat training course". Brain Research Bulletin . 48 (2): 203–9. doi : 10.1016/S0361-9230(98)00163-4 . PMID 10230711 . S2CID 27927524 .
- ↑ Mahoney CR, Castellani J, Kramer FM, Young A, Lieberman HR (November 2007). "Tyrosine supplementation mitigates working memory decrements during cold exposure" . Physiology & Behavior . 92 (4): 575–82. doi : 10.1016/j.physbeh.2007.05.003 . PMID 17585971 . S2CID 207372821 .
- ↑ Chinevere TD, Sawyer RD, Creer AR, Conlee RK, Parcell AC (November 2002). "Effects of L-tyrosine and carbohydrate ingestion on endurance exercise performance". Journal of Applied Physiology . 93 (5): 1590–7. doi : 10.1152/japplphysiol.00625.2001 . PMID 12381742 .
- ↑ Strüder HK, Hollmann W, Platen P, Donike M, Gotzmann A, Weber K (April 1998). "Influence of paroxetine, branched-chain amino acids and tyrosine on neuroendocrine system responses and fatigue in humans". Hormone and Metabolic Research . 30 (4): 188–94. doi : 10.1055/s-2007-978864 . PMID 9623632 .
- ↑ Thomas JR, Lockwood PA, Singh A, Deuster PA (November 1999). "Tyrosine improves working memory in a multitasking environment". Pharmacology, Biochemistry, and Behavior . 64 (3): 495–500. doi : 10.1016/S0091-3057(99)00094-5 . PMID 10548261 . S2CID 24717770 .
- ↑ Jung, Sophie E; Hase, Adrian; ann het Rot, Marije (2015). "Behavioral and cognitive effects of tyrosine intake in healthy human adults" . Pharmacology Biochemistry and Behavior . 133 : 1–6. doi : 10.1016/j.pbb.2015.03.008 . PMID 25797188 . S2CID 30331663 .
- 1 2 Lütke-Eversloh T, Santos CN, Stephanopoulos G (December 2007). "Perspectives of biotechnological production of L-tyrosine and its applications". Applied Microbiology and Biotechnology . 77 (4): 751–62. doi : 10.1007/s00253-007-1243-y . PMID 17968539 . S2CID 23088822 .
- ↑ Chavez-Bejar M, Baez-Viveros J, Martinez A, Bolivar F, Gosset G (2012). "Biotechnological production of L-tyrosine and derived compounds". Process Biochemistry . 47 (7): 1017–1026. doi : 10.1016/j.procbio.2012.04.005 .
External links
- Tyrosine MS Spectrum
- Tyrosine metabolism Archived 2019-07-26 at the Wayback Machine
- Phenylalanine and tyrosine biosynthesis
- Phenylalanine, Tyrosine, and tryptophan biosynthesis
- Tyrosine in the ChemIDplus database
| General topics | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| By properties |
|
||||||||||
| K → acetyl-CoA |
|
||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| G |
|
||||||||||||||||||||||||||||||||
| Other |
|
| catecholamines |
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| tryptophan → serotonin |
|
||||||||||
| serotonin → melatonin |
| D 1 -like |
|
||||||
|---|---|---|---|---|---|---|---|
| D 2 -like |
|
||||||
|
Receptor
( ligands ) |
|
||||
|---|---|---|---|---|---|
|
Transporter
( blockers ) |
|
||||
|
Enzyme
( inhibitors ) |
|
||||
| Others | |||||
|
| History of chocolate | |
|---|---|
| Theobroma species |
|
| Components | |
| Types | |
| Products |
|
| Processes | |
| Industry |
|
| Other topics | |
|
Authority control
: National
|
|---|