Indacaterol
Chemical compound
|
|

|
|
| Clinical data | |
|---|---|
| Trade names | Onbrez, Arcapta |
| AHFS / Drugs.com | International Drug Names |
| License data |
|
|
Pregnancy
category |
|
|
Routes of
administration |
Inhalation |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
|
|
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard ( EPA ) | |
| ECHA InfoCard |
100.218.577
|
| Chemical and physical data | |
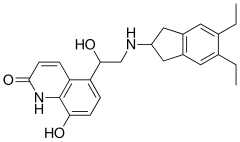
| Formula | C 24 H 28 N 2 O 3 |
| Molar mass | 392.499 g·mol −1 |
| 3D model ( JSmol ) | |
|
|
|
|
 N
N
 Y
(what is this?)
(verify)
Y
(what is this?)
(verify)
|
Indacaterol is an ultra- long-acting beta-adrenoceptor agonist [5] developed by Novartis . It needs to be taken only once a day, [6] unlike the related drugs formoterol and salmeterol . It is licensed only for the treatment of chronic obstructive pulmonary disease (COPD) (long-term data in patients with asthma are thus far lacking). It is delivered as an aerosol formulation through a dry powder inhaler .
Medical uses
A Cochrane review found benefit in lung function in people with COPD at least as good as that seen with twice-daily long-acting beta2-agonists. [7]
History
It was approved by the European Medicines Agency (EMA) under the brand name Onbrez Breezhaler on November 30, 2009, [8] and by the United States Food and Drug Administration (FDA), under the brand name Arcapta Neohaler, on July 1, 2011. [9] [10] In 2016, Novartis licensed its U.S. commercial rights for Arcapta Neohaler to Sunovion Pharmaceuticals . [11]
References
- ↑ "Arcapta Neohaler (indacaterol) inhalation powder Initial U.S. Approval: 2011" . DailyMed . 1 April 2020 . Retrieved 14 June 2021 .
- ↑ "Onbrez Breezhaler EPAR" . European Medicines Agency (EMA) . Retrieved 20 January 2021 .
- ↑ "Oslif Breezhaler EPAR" . European Medicines Agency (EMA) . Retrieved 20 January 2021 .
- ↑ "Hirobriz Breezhaler EPAR" . European Medicines Agency (EMA) . Retrieved 20 January 2021 .
- ↑ Cazzola M, Matera MG, Lötvall J (July 2005). "Ultra long-acting beta 2-agonists in development for asthma and chronic obstructive pulmonary disease". Expert Opin Investig Drugs . 14 (7): 775–83. doi : 10.1517/13543784.14.7.775 . PMID 16022567 . S2CID 11930383 .
- ↑ Beeh KM, Derom E, Kanniess F, Cameron R, Higgins M, van As A (May 2007). "Indacaterol, a novel inhaled beta2-agonist, provides sustained 24-h bronchodilation in asthma" . Eur. Respir. J . 29 (5): 871–8. doi : 10.1183/09031936.00060006 . PMID 17251236 .
- ↑ Geake, James B (2015). "Indacaterol, a once-daily beta2-agonist, versus twice-daily beta2-agonists or placebo for chronic obstructive pulmonary disease" . Reviews . 1 : CD010139. doi : 10.1002/14651858.CD010139.pub2 . PMC 6464646 . PMID 25575340 .
- ↑ European Public Assessment Report for Onbrez Breezhaler Archived 2010-01-16 at the Wayback Machine
- ↑ "FDA approves Arcapta Neohaler to treat chronic obstructive pulmonary disease" (Press release). U.S. Food and Drug Administration. 2011-07-01. Archived from the original on 2011-07-03 . Retrieved 2011-07-02 .
- ↑ "Drug Approval Package: Arcapta Neohaler (indacaterol maleate) NDA #022383" . U.S. Food and Drug Administration . 13 August 2013 . Retrieved 14 June 2021 .
- ↑ Faulkner, Sarah (22 December 2016). "Sunovion, Novartis ink licensing deal for inhaled COPD drugs" . Drug Delivery Business .
| Adrenergics , inhalants |
|
||||||||
|---|---|---|---|---|---|---|---|---|---|
| Glucocorticoids | |||||||||
|
Anticholinergics
/
muscarinic antagonist |
|||||||||
| Mast cell stabilizers | |||||||||
| Xanthines | |||||||||
| Eicosanoid inhibition |
|
||||||||
| Others/unknown | |||||||||
| Combination products |
|
||||||||
|