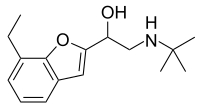
Bufuralol
Chemical compound
|
|
| Names | |
|---|---|
|
Preferred IUPAC name
2-(
tert
-Butylamino)-1-(7-ethyl-1-benzofuran-2-yl)ethan-1-ol
|
|
|
Other names
2-(
tert
-Butylamino)-1-(7-ethylbenzofuran-2-yl)ethan-1-ol
2-( tert -Butylamino)-1-(7-ethyl-1-benzofuran-2-yl)ethanol |
|
| Identifiers | |
|
3D model (
JSmol
)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard |
100.053.720
|
|
PubChem
CID
|
|
| UNII | |
|
CompTox Dashboard
(
EPA
)
|
|
|
|
|
|
| Properties | |
| C 16 H 23 N O 2 | |
| Molar mass | 261.365 g·mol −1 |
|
Except where otherwise noted, data are given for materials in their
standard state
(at 25
°C [77
°F], 100
kPa).
|
Bufuralol is a potent beta-adrenoceptor antagonist with partial agonist activity. [1] It is metabolized by CYP2D6 . [2]
Most beta blockers are aryloxypropanolamine-based. In this rare exception, the benzofuran oxygen is part of a ring instead of derived from the epichlorohydrin precursor.
References
- ↑ Pringle, TH; Francis, RJ; East, PB; Shanks, RG (1986). "Pharmacodynamic and pharmacokinetic studies on bufuralol in man" . British Journal of Clinical Pharmacology . 22 (5): 527–34. doi : 10.1111/j.1365-2125.1986.tb02931.x . PMC 1401192 . PMID 2878678 .
- ↑ Flockhart DA (2007). "Drug Interactions: Cytochrome P 450 Drug Interaction Table" . Indiana University School of Medicine . Retrieved on July 2011

|
This drug article relating to the cardiovascular system is a stub . You can help Wikipedia by expanding it . |
