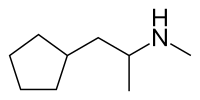
Cyclopentamine
Decongestant and stimulant drug
|
|

|
|
| Clinical data | |
|---|---|
| Other names | N ,α-dimethyl-cyclopenaneethylamine |
|
Routes of
administration |
Topical ( nasal spray ) |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
|
|
| CAS Number |
|
| PubChem CID | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEMBL | |
| Chemical and physical data | |
| Formula | C 9 H 19 N |
| Molar mass | 141.258 g·mol −1 |
| 3D model ( JSmol ) | |
| Chirality | Racemic mixture |
| Boiling point | 171 °C (340 °F) |
|
|
|
|
 N
N
 Y
(what is this?)
(verify)
Y
(what is this?)
(verify)
|
Cyclopentamine (trade names Clopane , Cyclonarol , Cyclosal , Cyklosan , Nazett , Sinos , among others) is a sympathomimetic alkylamine , classified as a vasoconstrictor . Cyclopentamine was indicated in the past as an over-the-counter (OTC) medication for use as a nasal decongestant , notably in Europe and Australia , but has now been largely discontinued.
Pharmacology
Cyclopentamine acts as a releasing agent of the catecholamine neurotransmitters norepinephrine (noradrenaline), epinephrine (adrenaline), and dopamine . [1] Its effects on norepinephrine and epinephrine mediate its decongestant effects, while its effects on all three neurotransmitters are responsible for its stimulant properties. When ingested orally in sufficient quantities , cyclopentamine produces similar effects to amphetamine , methamphetamine, and propylhexedrine. [2] [3]
Chemistry
Cyclopentamine is the cyclopentane homolog of propylhexedrine , differing only in terms of the contracted ring size of a cyclopentane, containing one —CH 2 — unit less than the cyclohexyl group.
In terms of the acyclic part of the molecule, both cyclopentamine and propylhexedrine are the same as methamphetamine , all three molecules containing the 2-methylaminopropyl side-chain. The difference between them is that whereas methamphetamine is an aromatic molecule containing a phenyl group , cyclopentamine and propylhexedrine are entirely aliphatic and contain no delocalized electrons at all. The effect that this has on potency is that the reduced alicyclic -alkylamines are weaker than unsaturated (meth)amphetamine. [ citation needed ]
See also
- Amphetamine
- Cypenamine (which is trans -2-phenylcyclopentylamine)
- Methamphetamine
- Propylhexedrine (also known as cyclohexylisopropylmethylamine)
- Tranylcypromine (which is trans -2-phenylcyclopropylamine)
- Methiopropamine
References
- ↑ Schmidt JL, Fleming WW (July 1964). "A Nonsympathomimetic Effect of Cyclopentamine and Beta-Mercaptoethylamine in the Rabbit Ileum". The Journal of Pharmacology and Experimental Therapeutics . 145 : 83–6. PMID 14209515 .
- ↑ Ghouri MS, Haley TJ (July 1969). "In vitro evaluation of a series of sympathomimetic amines and the beta-adrenergic blocking properties of cyclopentamine". Journal of Pharmaceutical Sciences . 58 (7): 882–4. doi : 10.1002/jps.2600580722 . PMID 4390216 .
- ↑ Marley E, Stephenson JD (August 1971). "Actions of dexamphetamine and amphetamine-like amines in chickens with brain transections" . British Journal of Pharmacology . 42 (4): 522–42. doi : 10.1111/j.1476-5381.1971.tb07138.x . PMC 1665761 . PMID 5116035 .
|
Decongestants
and other nasal preparations (
R01
)
|
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Topical |
|
||||||||||
|
Systemic use:
Sympathomimetics |
|||||||||||
|