Cypenamine
Chemical compound
|
|
| Clinical data | |
|---|---|
| Other names | 2-Phenylcyclopentanamine |
| ATC code |
|
| Identifiers | |
|
|
| CAS Number | |
| PubChem CID | |
| ChemSpider |
|
| UNII | |
| ChEMBL | |
| CompTox Dashboard ( EPA ) | |
| Chemical and physical data | |
| Formula | C 11 H 15 N |
| Molar mass | 161.248 g·mol −1 |
| 3D model ( JSmol ) | |
| Chirality | Racemic mixture |
|
|
|
|
| (verify) |
Cypenamine ( INN , BAN ), or cypenamine hydrochloride ( USAN ), also known as 2-phenylcyclopentylamine , is a psychostimulant drug which was developed by a group at the William S. Merrell Chemical Company in the 1940s. [1] [2] It is currently known only in scientific research and has never been developed for market use . Cypenamine is currently legal throughout the entire world , and though its chemical structure has a vague similarity to certain controlled stimulants like fencamfamine , it is likely that it is too distant for it to be considered an illicit analogue under the United States Federal Analogue Act of the Controlled Substances Act .
Chemistry
Stereochemistry
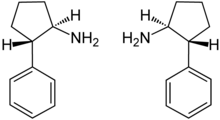
2-Phenylcyclopentan-1-amine is a compound with two stereocenters . Thus, the following two enantiomeric pairs may exist:
- (1 RS ,2 SR )- trans -2-phenylcyclopentan-1-amine
- (1 RS ,2 RS )- cis -2-phenylcyclopentan-1-amine
The racemate (±)- trans -2-phenylcyclopentan-1-amine [1:1 mixture of (1 R ,2 S )- trans -2-phenylcyclopentan-1-amine (box, left) and (1 S ,2 R )- trans -2-phenylcyclopentan-1-amine (box, right)] is the active ingredient of cypenamine. [3] Furthermore, the kinetic resolution of (±)- trans -2-phenylcyclopentan-1-amine by lipase B from Candida antarctica may be effectively performed by an aminolysis reaction. [3]
Racemic cis -2-phenylcyclopentan-1-amine [1:1 mixture of (1 R ,2 R )- cis -2-phenylcyclopentan-1-amine and (1 S ,2 S )- cis -2-phenylcyclopentan-1-amine] has found no pharmacological application. [ citation needed ]
Homology
Cypenamine is a homolog of tranylcypromine , containing an expanded alicyclic ring that is two methylene units larger than the highly strained/reactive cyclopropane. The cyclohexane homologue has been reported, although the LD 50 s were all less than for plain amphetamine, it was still a functional stimulant. [ citation needed ]
See also
References
- ↑ US patent 2520516 , van Zoeren GJ, "Cyclic Amines and Method of Making Them", issued 1950-08-29
- ↑ Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies . Springer. pp. 966–. ISBN 978-1-4757-2085-3 .
- 1 2 González-Sabín J, Gotor V, Rebolledo F (2004). "Kinetic resolution of (±)-trans- und (±)-cis-phenylcyclopentanamine by CALB-catalyzed aminolysis of esters: The key role of the leaving group". Tetrahedron:Asymmetry . 15 (3): 481–488. doi : 10.1016/j.tetasy.2003.11.013 .
