2,5-Dimethoxy-4-iodoamphetamine
Chemical compound
|
|
|
|
| Names | |
|---|---|
|
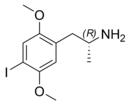
Preferred IUPAC name
1-(4-Iodo-2,5-dimethoxyphenyl)propan-2-amine
|
|
| Identifiers | |
|
|
|
3D model (
JSmol
)
|
|
| ChEMBL | |
| ChemSpider |
|
|
PubChem
CID
|
|
| UNII | |
|
CompTox Dashboard
(
EPA
)
|
|
|
|
|
|
| Properties | |
| C 11 H 16 INO 2 | |
| Molar mass | 321.1558 g/mol |
| Melting point | 201.5 °C (394.7 °F; 474.6 K) ( hydrochloride ) |
| 10 mg/mL [1] | |
|
Except where otherwise noted, data are given for materials in their
standard state
(at 25
°C [77
°F], 100
kPa).
|
2,5-Dimethoxy-4-iodoamphetamine ( DOI ) is a psychedelic drug and a substituted amphetamine . Unlike many other substituted amphetamines, however, it is not primarily a stimulant . [2] DOI has a stereocenter and R -(−)-DOI is the more active stereoisomer . In neuroscience research, [ 125 I ]- R -(−)-DOI is used as a radioligand and indicator of the presence of 5-HT 2A serotonin receptors . DOI's effects have been compared to LSD , although there are differences that experienced users can distinguish. Besides the longer duration, the trip tends to be more energetic than an LSD trip, with more body load and a different subjective visual experience. The after effects include residual stimulation and difficulty sleeping, which, depending on the dose, may persist for days. [2] While rare, it is sometimes sold as a substitute for LSD, or even sold falsely as LSD, which may be dangerous because DOI does not have the same established safety profile as LSD. [3]
Research
Research [4] suggests that administration of ( R )-DOI blocks pulmonary inflammation, mucus hyper-production, airway hyper-responsiveness and turns off key genes in in-lung immune response. These effects block the development of allergic asthma in a mouse model.
Several 5-HT 2A agonist hallucinogens including ( R )-2,5-dimethoxy-4-iodoamphetamine DOI, TCB-2 , LSD and LA-SS-Az have unexpectedly also been found to act as potent inhibitors of TNF , with DOI being the most active, showing TNF inhibition in the picomolar range, an order of magnitude more potent than its action as a hallucinogen. [5] [6] [7]
Pharmacology
| Receptor | K i (racemic DOI) [8] [ failed verification ] | K i ( R -DOI) [8] [ failed verification ] | K i ( S -DOI) [8] [ failed verification ] | Intrinsic activity [9] |
|---|---|---|---|---|
| 5-HT 1A | 2355 nM | 3843 nM | ND | ND |
| 5-HT 1B | 1261 nM | ND | ND | ND |
| 5-HT 1D | 1241.3 nM | ND | ND | ND |
| 5-HT 1E | 2970 nM | ND | ND | ND |
| 5-HT 1F | 2125.44 nM | ND | ND | ND |
| 5-HT 2A | 0.68 nM | 0.65 nM | 0.65 nM | Partial agonist. |
| 5-HT 2B | 20.03 nM | 53.70318 nM | 28.183829 nM | Partial agonist/full agonist |
| 5-HT 2C | 2.38 nM | 5.370318 nM | 8.317638 nM | Full agonist when coupled to phospholipase A . Partial agonist (intrinsic efficacy = 53%), when coupled to phospholipase C . |
| 5-HT 5A | 1000 nM | ND | ND | ND |
| 5-HT 6 | >10000 nM | ND | ND | ND |
DOI is a 5-HT 2A , 5-HT 2B and 5-HT 2C receptor agonist. [9]
DOI has been shown to be an extremely potent inhibitor of tumour necrosis factor-alpha inflammation at picomolar concentrations in cell studies. TNF-alpha is an important target for research into degenerative conditions such as rheumatoid arthritis and Alzheimer's disease , where the disease process involves tissue damage through chronic inflammation . This could make DOI and other 5-HT 2A agonists an entirely new area for development of novel treatments for these conditions. [10]
DOI has also been shown to induce rapid growth and reorganization of dendritic spines and synaptic connections with other neurons , processes known to underlie neuroplasticity . [11]
History
DOI was first synthesized by Alexander Shulgin . [2] The radioactive iodine-125 form of DOI for PET imaging was first developed in the lab of David E. Nichols .
In January 2007, British police reported that three young men had fallen ill, reportedly, after taking DOI at a rave in Biggleswade , near Milton Keynes , and warned others who had taken it to seek medical attention. This would appear to be the first indication that DOI has found more widespread use as a recreational drug in the UK. [12]
Legal status
Australia
The Standard for the Uniform Scheduling of Medicines and Poisons (SUSMP) of Australia does not list DOI as a prohibited substance. [13]
Canada
Listed as a Schedule 1 [14] as it is an analogue of amphetamine. [15] The CDSA was updated as a result of the Safe Streets and Communities Act , changing amphetamines from Schedule 3 to Schedule 1 . [16]
Denmark
Illegal since 8 April 2007. [17]
Sweden
Sveriges riksdag added DOI to schedule I ( "substances, plant materials and fungi which normally do not have medical use" ) as narcotics in Sweden as of August 30, 2007, published by Medical Products Agency in their regulation LVFS 2007:10 listed as DOI, 4-jod-2,5-dimetoxi-amfetamin . [18]
United States
DOI is not scheduled in the United States, [19] but it is likely that DOI would be considered an analog (of DOB ), in which case, sales or possession could be prosecuted under the Federal Analogue Act . DOI is regularly used in animal and in vitro research. [20] Scheduling DOI could cause problems for medical researchers. [21]
US State of Florida
DOI is a Schedule I controlled substance in the state of Florida. [22]
See also
References
- ↑ "D101 DOI hydrochloride ≥98% (HPLC), solid" . Retrieved 13 April 2008 .
-
1
2
3
Shulgin, A; Shulgin, A (1990).
"#67 DOI"
.
PiHKAL: A Chemical Love Story
. Transform Press.
ISBN
978-0963009609
. Archived from the original on 2014-10-27
. Retrieved
2014-12-17
.
{{ cite book }}: CS1 maint: bot: original URL status unknown ( link ) - ↑ "LSD Blotter Acid Mimics (Actually containing 4-IODO-2,5-DIMETHOXYAMPHETAMINE (DOI) and 4-CHLORO-2,5-DIMETHOXYAMPHETAMINE (DOC)) in Lantana, Florida" . DEA Microgram Bulletin . Washington, DC: Office of Forensic Sciences, Drug Enforcement Administration. June 2008. Archived from the original on 2009-02-04 . Retrieved 12 February 2009 .
- ↑ "LSU Health New Orleans research finds psychedelic drug prevents asthma development in mice" . EurekAlert! .
- ↑ Miller KJ, Gonzalez HA (December 1998). "Serotonin 5-HT2A receptor activation inhibits cytokine-stimulated inducible nitric oxide synthase in C6 glioma cells". Ann. N. Y. Acad. Sci . 861 (1): 169–73. Bibcode : 1998NYASA.861..169M . doi : 10.1111/j.1749-6632.1998.tb10188.x . PMID 9928254 . S2CID 23264746 .
- ↑ Yu B, Becnel J, Zerfaoui M, Rohatgi R, Boulares AH, Nichols CD (November 2008). "Serotonin 5-hydroxytryptamine(2A) receptor activation suppresses tumor necrosis factor-alpha-induced inflammation with extraordinary potency". J. Pharmacol. Exp. Ther . 327 (2): 316–23. doi : 10.1124/jpet.108.143461 . PMID 18708586 . S2CID 25374241 .
- ↑ Pelletier M, Siegel RM (December 2009). "Wishing away inflammation? New links between serotonin and TNF signaling" . Mol. Interv . 9 (6): 299–301. doi : 10.1124/mi.9.6.5 . PMC 2861806 . PMID 20048135 .
- 1 2 3 Roth, BL ; Driscol, J (12 January 2011). "PDSP K i Database" . Psychoactive Drug Screening Program (PDSP) . University of North Carolina at Chapel Hill and the United States National Institute of Mental Health. Archived from the original on 8 November 2013 . Retrieved 4 March 2014 .
- 1 2 Canal, CE; Morgan, D (July 2012). "Head-twitch response in rodents induced by the hallucinogen 2,5-dimethoxy-4-iodoamphetamine: a comprehensive history, a re-evaluation of mechanisms, and its utility as a model" . Drug Testing and Analysis . 4 (7–8): 556–576. doi : 10.1002/dta.1333 . PMC 3722587 . PMID 22517680 .
- ↑ Yu, B; Becnel, J; Zerfaoui, M; Rohatgi, R; Boulares, AH; Nichols, CD (2008). "Serotonin 5-Hydroxytryptamine2A Receptor Activation Suppresses Tumor Necrosis Factor-α-Induced Inflammation with Extraordinary Potency". Journal of Pharmacology and Experimental Therapeutics . 327 (2): 316–323. doi : 10.1124/jpet.108.143461 . PMID 18708586 . S2CID 25374241 .
- ↑ Jones, KA; Srivastava, DP; Allen, JA; Strachan, RT; Roth, BL; Penzes, P (17 November 2009). "Rapid modulation of spine morphology by the 5-HT2A serotonin receptor through kalirin-7 signaling" . Proceedings of the National Academy of Sciences . 106 (46): 19575–19580. Bibcode : 2009PNAS..10619575J . doi : 10.1073/pnas.0905884106 . PMC 2780750 . PMID 19889983 .
- ↑ "New drug alert as three taken ill" . BBC News . 29 January 2007.
- ↑ Gill, A (22 July 2013). "POISONS STANDARD 2013" (PDF) . Therapeutic Goods Administration. Australian Government Department of Health and Ageing . Retrieved 4 March 2014 .
- ↑ "Controlled Drugs and Substances Act : Legislative history · Schedule I · Section 19: Tramadol [ Proposed ] ; Amphetamines" . isomerdesign.com . Archived from the original on 2022-03-31 . Retrieved 2012-11-27 .
- ↑ "Controlled Drugs and Substances Act : Definitions and Interpretations" . isomerdesign.com .
- ↑ (in English)
- ↑ "Erowid DOC Vault : Legal Status" . www.erowid.org .
- ↑ "Läkemedelsverkets föreskrifter - LVFS och HSLF-FS | Läkemedelsverket" (PDF) .
- ↑ "PART 1308 - Section 1308.11 Schedule I" . www.deadiversion.usdoj.gov . Archived from the original on 2009-08-27 . Retrieved 2014-12-17 .
- ↑ Mario de la Fuente Revenga; Bohan Zhu; Christopher A. Guevara; George W. Huntley; Chang Lu; Javier González-Maeso (2021). "Prolonged epigenomic and synaptic plasticity alterations following single exposure to a psychedelic in mice" . Cell Reports . 37 (3): 109836. doi : 10.1016/j.celrep.2021.109836 . PMC 8582597 . PMID 34686347 .
- ↑ "Schedules of Controlled Substances: Placement of 2,5-dimethoxy-4-iodoamphetamine (DOI) and 2,5-dimethoxy-4-chloroamphetamine (DOC) in Schedule I" . www.regulations.gov .
- ↑ "Statutes & Constitution :View Statutes : Online Sunshine" . leg.state.fl.us .
External links
| 5-HT 1 |
|
||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 5-HT 2 |
|
||||||||||||||||||||||||
| 5-HT 3 – 7 |
|
||||||||||||||||||||||||
| Phenethylamines |
|
|---|---|
| Amphetamines |
|
| Phentermines |
|
| Cathinones |
|
| Phenylisobutylamines | |
| Phenylalkylpyrrolidines | |
|
Catecholamines
(and close relatives) |
|
| Miscellaneous |
|


