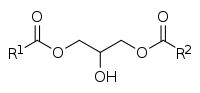
Diglyceride
Type of fat derived from glycerol and two fatty acids
A diglyceride , or diacylglycerol ( DAG ), is a glyceride consisting of two fatty acid chains covalently bonded to a glycerol molecule through ester linkages. [1] Two possible forms exist, 1,2-diacylglycerols and 1,3-diacylglycerols. DAGs can act as surfactants and are commonly used as emulsifiers in processed foods. DAG-enriched oil (particularly 1,3-DAG) has been investigated extensively as a fat substitute due to its ability to suppress the accumulation of body fat; [2] [3] with total annual sales of approximately USD 200 million in Japan since its introduction in the late 1990s till 2009. [2]
Production
Diglycerides are a minor component of many seed oils and are normally present at ~1–6%; or in the case of cottonseed oil as much as 10%. [4] Industrial production is primarily achieved by a glycerolysis reaction between triglycerides and glycerol. The raw materials for this may be either vegetable oils or animal fats . [5]
Food additive
Diglycerides, generally in a mix with monoglycerides ( E471 ), are common food additives largely used as emulsifiers . The values given in the nutritional labels for total fat, saturated fat, and trans fat do not include those present in mono- and diglycerides. [ citation needed ] They often are included in bakery products, beverages, ice cream , peanut butter , chewing gum , shortening , whipped toppings, margarine , confections, and some snack products, such as Pringles .
Biological functions
Protein kinase C activation

In biochemical signaling, diacylglycerol functions as a second messenger signaling lipid , and is a product of the hydrolysis of the phospholipid phosphatidylinositol 4,5-bisphosphate (PIP 2 ) by the enzyme phospholipase C (PLC) (a membrane -bound enzyme) that, through the same reaction, produces inositol trisphosphate (IP 3 ). Although inositol trisphosphate diffuses into the cytosol , diacylglycerol remains within the plasma membrane , due to its hydrophobic properties. IP 3 stimulates the release of calcium ions from the smooth endoplasmic reticulum , whereas DAG is a physiological activator of protein kinase C (PKC). The production of DAG in the membrane facilitates translocation of PKC from the cytosol to the plasma membrane .
Munc13 activation
Diacylglycerol has been shown to exert some of its excitatory actions on vesicle release through interactions with the presynaptic priming protein family Munc13 . Binding of DAG to the C1 domain of Munc13 increases the fusion competence of synaptic vesicles resulting in potentiated release.
Diacylglycerol can be mimicked by the tumor-promoting compounds phorbol esters . [6]
Other
In addition to activating PKC, diacylglycerol has a number of other functions in the cell :
- a source for prostaglandins
- a precursor of the endocannabinoid 2-arachidonoylglycerol
- an activator of a subfamily of transient receptor potential canonical (TRPC) cation channels, TRPC3/6/7.
Metabolism
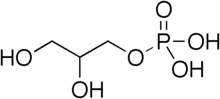
Synthesis of diacylglycerol begins with glycerol-3-phosphate , which is derived primarily from dihydroxyacetone phosphate , a product of glycolysis (usually in the cytoplasm of liver or adipose tissue cells). Glycerol-3-phosphate is first acylated with acyl-coenzyme A (acyl-CoA) to form lysophosphatidic acid , which is then acylated with another molecule of acyl-CoA to yield phosphatidic acid . Phosphatidic acid is then de-phosphorylated to form diacylglycerol.
Dietary fat is mainly composed of triglycerides . Because triglycerides cannot be absorbed by the digestive system, triglycerides must first be enzymatically digested into monoacylglycerol , diacylglycerol, or free fatty acids. Diacylglycerol is a precursor to triacylglycerol (triglyceride), which is formed in the addition of a third fatty acid to the diacylglycerol under the catalysis of diglyceride acyltransferase .
Since diacylglycerol is synthesized via phosphatidic acid, it will usually contain a saturated fatty acid at the C-1 position on the glycerol moiety and an unsaturated fatty acid at the C-2 position. [7]
Diacylglycerol can be phosphorylated to phosphatidic acid by diacylglycerol kinase .
Insulin resistance
Activation of PKC-θ by diacylglycerol may cause insulin resistance in muscle by decreasing IRS1 -associated PI3K activity. [8] Similarly, activation of PKCε by diacyglycerol may cause insulin resistance in the liver. [8] [9]
See also
References
- ↑ IUPAC , Compendium of Chemical Terminology , 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006 – ) " glycerides ". doi : 10.1351/goldbook.G02647
- 1 2 Phuah, Eng-Tong; Tang, Teck-Kim; Lee, Yee-Ying; et al. (2015). "Review on the Current State of Diacylglycerol Production Using Enzymatic Approach" (PDF) . Food and Bioprocess Technology . 8 (6): 1169–1186. doi : 10.1007/s11947-015-1505-0 . ISSN 1935-5130 . S2CID 84353775 .
- ↑ Lo, Seong-Koon; Tan, Chin-Ping; Long, Kamariah; et al. (2008). "Diacylglycerol Oil—Properties, Processes and Products: A Review" (PDF) . Food and Bioprocess Technology . 1 (3): 223–233. doi : 10.1007/s11947-007-0049-3 . ISSN 1935-5130 . S2CID 86604260 .
- ↑ Flickinger, Brent D.; Matsuo, Noboru (February 2003). "Nutritional characteristics of DAG oil". Lipids . 38 (2): 129–132. doi : 10.1007/s11745-003-1042-8 . PMID 12733744 . S2CID 4061326 .
- ↑ Sonntag, Norman O. V. (1982). "Glycerolysis of fats and methyl esters — Status, review and critique". Journal of the American Oil Chemists' Society . 59 (10): 795A–802A. doi : 10.1007/BF02634442 . ISSN 0003-021X . S2CID 84808531 .
- ↑ Blumberg, Peter M. (1988). "Protein Kinase C as the Receptor for the Phorbol Ester Tumor Promoters: Sixth Rhoads Memorial Award Lecture" . Cancer Research . 48 (1): 1–8. PMID 3275491 .
- ↑ Berg J, Tymoczko JL, Stryer L (2006). Biochemistry (6th ed.). San Francisco: W. H. Freeman. ISBN 0-7167-8724-5 . [ page needed ]
- 1 2 Erion DM, Shulman GI (2010). "Diacylglycerol-mediated insulin resistance" . Nature Medicine . 16 (4): 400–402. doi : 10.1038/nm0410-400 . PMC 3730126 . PMID 20376053 .
- ↑ Petersen MC, Madiraju AK, Gassaway BM, et al. (2016). "Insulin receptor Thr1160 phosphorylation mediates lipid-induced hepatic insulin resistance" . Journal of Clinical Investigation . 126 (11): 4361–4371. doi : 10.1172/JCI86013 . PMC 5096902 . PMID 27760050 .
|
Types of
lipids
|
|
|---|---|
| General |
|
| Geometry | |
| Eicosanoids | |
| Fatty acids | |
| Glycerides | |
| Phospholipids | |
| Sphingolipids | |
| Steroids |
|
Receptor
( ligands ) |
|
||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Enzyme
( inhibitors ) |
|
||||||||||||||||||||||||||
| Others | |||||||||||||||||||||||||||
|
Receptor
( ligands ) |
|
||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Enzyme
( inhibitors ) |
|
||||||||||||||||
| Others | |||||||||||||||||
| TRPA |
|
||||
|---|---|---|---|---|---|
| TRPC |
|
||||
| TRPM |
|
||||
| TRPML |
|
||||
| TRPP |
|
||||
| TRPV |
|
||||
|
See also:
Receptor/signaling modulators
•
Ion channel modulators
|