Isoxicam
Chemical compound
|
|
| Clinical data | |
|---|---|
| Trade names | Maxicam |
| ATC code | |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEBI | |
| CompTox Dashboard ( EPA ) | |
| ECHA InfoCard |
100.047.334
|
| Chemical and physical data | |
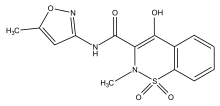
| Formula | C 14 H 13 N 3 O 5 S |
| Molar mass | 335.33 g·mol −1 |
| 3D model ( JSmol ) | |
|
Isoxicam is a nonsteroidal anti-inflammatory drug (NSAID) that was taken or applied to reduce inflammation and as an analgesic reducing pain in certain conditions. The drug was introduced in 1983 by the Warner-Lambert Company . Isoxicam is a chemical analog of piroxicam (Feldene) which has a pyridine ring in lieu of an isoxazole ring. In 1985 Isoxicam was withdrawn from the French market, due to adverse effects, namely Toxic Epidermal Necrolysis ( Lyell syndrome ) resulting in death. Although these serious side effects were observed only in France, the drug was withdrawn worldwide. [1] [2]
References
- ↑ Consolidated List of products whose consumption and/or sale have been banned, withdrawn, severely restricted or not approved by Governments , United Nations, 2003, p. 123 link to 2005 ed
- ↑ Fung M, Thornton A, Mybeck K, Wu JH, Hornbuckle K, Muniz E (January 2001). "Evaluation of the Characteristics of Safety Withdrawal of Prescription Drugs from Worldwide Pharmaceutical Markets-1960 to 1999". Therapeutic Innovation & Regulatory Science . 35 (1): 293–317. doi : 10.1177/009286150103500134 . S2CID 73036562 .
|
pyrazolones
/
pyrazolidines |
|
|---|---|
| salicylates | |
|
acetic acid
derivatives
and related substances |
|
| oxicams | |
|
propionic acid
derivatives (profens) |
|
|
n
-arylanthranilic
acids ( fenamates ) |
|
|
COX-2 inhibitors
(coxibs) |
|
| other | |
|
NSAID
combinations |
|
|
Key:
underline
indicates initially developed first-in-class compound of specific group;
#
WHO-Essential Medicines
;
†
withdrawn drugs
;
‡
veterinary use
.
|
|





