Selenium dioxide
Chemical compound

|
|
|
|

|
|
| Names | |
|---|---|
|
Other names
Selenium(IV) oxide
Selenous anhydride |
|
| Identifiers | |
|
3D model (
JSmol
)
|
|
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard |
100.028.358
|
| EC Number |
|
|
PubChem
CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 3283 |
|
CompTox Dashboard
(
EPA
)
|
|
|
|
|
|
| Properties | |
| SeO 2 | |
| Molar mass | 110.96 g/mol |
| Appearance | White crystals, turn slightly pink with trace decomposition [1] |
| Odor | rotten radishes |
| Density | 3.954 g/cm 3 , solid |
| Melting point | 340 °C (644 °F; 613 K) (sealed tube) |
| Boiling point | 350 °C (662 °F; 623 K) subl. |
|
38.4 g/100 mL (20 °C)
39.5 g/100 ml (25 °C) 82.5 g/100 mL (65 °C) |
|
| Solubility | soluble in benzene |
| Solubility in ethanol | 6.7 g/100 mL (15 °C) |
| Solubility in acetone | 4.4 g/100 mL (15 °C) |
| Solubility in acetic acid | 1.11 g/100 mL (14 °C) |
| Solubility in methanol | 10.16 g/100 mL (12 °C) |
| Vapor pressure | 1.65 kPa (70 °C) |
| Acidity (p K a ) | 2.62; 8.32 |
|
Magnetic susceptibility
(
χ
)
|
− 27.2·10 −6 cm 3 /mol |
|
Refractive index
(
n
D
)
|
> 1.76 |
| Structure | |
| see text | |
| trigonal (Se) | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
|
Main hazards
|
Toxic by ingestion and inhalation [2] |
| GHS labelling : | |



|
|
| Danger | |
| H301 , H331 , H373 , H410 | |
| P260 , P261 , P264 , P270 , P271 , P273 , P301+P310 , P304+P340 , P311 , P314 , P321 , P330 , P391 , P403+P233 , P405 , P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
|
LC
Lo
(
lowest published
)
|
5890 mg/m
3
(rabbit, 20 min)
6590 mg/m 3 (goat, 10 min) 6590 mg/m 3 (sheep, 10 min) [3] |
| Safety data sheet (SDS) | ICSC 0946 |
| Related compounds | |
|
Other
anions
|
Selenium disulfide |
|
Other
cations
|
Sulfur dioxide
Tellurium dioxide |
| Selenium trioxide | |
|
Related compounds
|
Selenous acid |
|
Except where otherwise noted, data are given for materials in their
standard state
(at 25
°C [77
°F], 100
kPa).
|
Selenium dioxide is the chemical compound with the formula SeO 2 . This colorless solid is one of the most frequently encountered compounds of selenium .
Properties
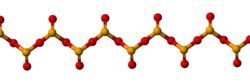
Solid SeO 2 is a one-dimensional polymer , the chain consisting of alternating selenium and oxygen atoms. Each Se atom is pyramidal and bears a terminal oxide group. The bridging Se-O bond lengths are 179 pm and the terminal Se-O distance is 162 pm. [4] The relative stereochemistry at Se alternates along the polymer chain ( syndiotactic ). In the gas phase selenium dioxide is present as dimers and other oligomeric species, at higher temperatures it is monomeric. [5] The monomeric form adopts a bent structure very similar to that of sulfur dioxide with a bond length of 161 pm. [5] The dimeric form has been isolated in a low temperature argon matrix and vibrational spectra indicate that it has a centrosymmetric chair form. [4] Dissolution of SeO 2 in selenium oxydichloride give the trimer [Se(O)O] 3 . [5] Monomeric SeO 2 is a polar molecule, with the dipole moment of 2.62 D [6] pointed from the midpoint of the two oxygen atoms to the selenium atom.
The solid sublimes readily. At very low concentrations the vapour has a revolting odour, resembling decayed horseradishes. At higher concentrations the vapour has an odour resembling horseradish sauce and can burn the nose and throat on inhalation. Whereas SO 2 tends to be molecular and SeO 2 is a one-dimensional chain, TeO 2 is a cross-linked polymer. [4]
SeO
2
is considered an
acidic oxide
: it dissolves in water to form
selenous acid
.
[5]
Often the terms
selenous acid
and
selenium dioxide
are used interchangeably. It reacts with base to form
selenite
salts
containing the
SeO
2−
3
anion. For example, reaction with
sodium hydroxide
produces
sodium selenite
:
- SeO 2 + 2 NaOH → Na 2 SeO 3 + H 2 O
Preparation
Selenium dioxide is prepared by oxidation of selenium by burning in air or by reaction with nitric acid or hydrogen peroxide , but perhaps the most convenient preparation is by the dehydration of selenous acid .
- 2 H 2 O 2 + Se → SeO 2 + 2 H 2 O
- 3 Se + 4 HNO 3 + H 2 O → 3 H 2 SeO 3 + 4 NO
- H 2 SeO 3 ⇌ SeO 2 + H 2 O
Occurrence
The natural form of selenium dioxide, downeyite, is a very rare mineral. It is only found at a small number of burning coal banks , where it forms around vents created from escaping gasses. [7]
Uses
Organic synthesis
SeO 2 is an important reagent in organic synthesis . Oxidation of paraldehyde (acetaldehyde trimer) with SeO 2 gives glyoxal [8] and the oxidation of cyclohexanone gives 1,2-Cyclohexanedione . [9] The selenium starting material is reduced to selenium, and precipitates as a red amorphous solid which can easily be filtered off. [9] This type of reaction is called a Riley oxidation . It is also renowned as a reagent for "allylic" oxidation, [10] a reaction that entails the following conversion

This can be described more generally as;
- R 2 C=CR'-CHR" 2 + [O] → R 2 C=CR'-C(OH)R" 2
where R, R', R" may be alkyl or aryl substituents.
Selenium dioxide can also be used to synthesize 1,2,3-selenadiazoles from acylated hydrazone derivatives. [11]
As a colorant
Selenium dioxide imparts a red colour to glass . It is used in small quantities to counteract the colour due to iron impurities and so to create (apparently) colourless glass. In larger quantities, it gives a deep ruby red colour.
Selenium dioxide is the active ingredient in some cold-bluing solutions.
It was also used as a toner in photographic developing .
Safety
Selenium is an essential element , but ingestion of more than 5 mg/day leads to nonspecific symptoms . [12]
References
- ↑ "Safety data sheet: Selenium dioxide" (PDF) . integrachem.com . 27 March 2015 . Retrieved 2022-12-02 .
- ↑ "Selenium dioxide safety and hazards" . pubchem.ncbi.nlm.nih.gov . Retrieved 2022-12-02 .
- ↑ "Selenium compounds (as Se)" . Immediately Dangerous to Life or Health Concentrations (IDLH) . National Institute for Occupational Safety and Health (NIOSH).
- 1 2 3 Handbook of Chalcogen Chemistry: New Perspectives in Sulfur, Selenium and Tellurium, Franceso A. Devillanova, Royal Society of Chemistry, 2007, ISBN 9780854043668
- 1 2 3 4 Holleman, Arnold Frederik; Wiberg, Egon (2001), Wiberg, Nils (ed.), Inorganic Chemistry , translated by Eagleson, Mary; Brewer, William, San Diego/Berlin: Academic Press/De Gruyter, ISBN 0-12-352651-5
- ↑ Takeo, Harutoshi; Hirota, Eizi; Morino, Yonezo (1972). "Third-order potential constants and dipole moment of SeO2 by microwave spectroscopy". Journal of Molecular Spectroscopy . 41 (2): 420–422. Bibcode : 1972JMoSp..41..420T . doi : 10.1016/0022-2852(72)90216-0 . ISSN 0022-2852 .
- ↑ Finkelman, Robert B.; Mrose, Mary E. (1977). "Downeyite, the first verified natural occurrence of SeO 2 " (PDF) . American Mineralogist . 62 : 316–320.
-
↑
Ronzio, A. R.; Waugh, T. D. (1955).
"Glyoxal Bisulfite"
.
Organic Syntheses
.
{{ cite journal }}: CS1 maint: multiple names: authors list ( link ) ; Collective Volume , vol. 3, p. 438 -
1
2
Hach, C. C. Banks, C. V.; Diehl, H. (1963).
"1,2-Cyclohexanedione Dioxime"
.
Organic Syntheses
.
{{ cite journal }}: CS1 maint: multiple names: authors list ( link ) ; Collective Volume , vol. 4, p. 229 -
↑
Coxon, J. M.; Dansted, E.;
Hartshorn, M. P.
(1988).
"Allylic Oxidation with Hydrogen Peroxide–Selenium Dioxide: trans-Pinocarveol"
.
Organic Syntheses
.
{{ cite journal }}: CS1 maint: multiple names: authors list ( link ) ; Collective Volume , vol. 6, p. 946 - ↑ Lalezari, Iraj ; Shafiee, Abbas; Yalpani, Mohamed (1969). "A novel synthesis of selenium heterocycles: substituted 1,2,3-selenadiazoles". Tetrahedron Letters . 10 (58): 5105–5106. doi : 10.1016/S0040-4039(01)88895-X .
- ↑ Bernd E. Langner "Selenium and Selenium Compounds" in Ullmann's Encyclopedia of Industrial Chemistry, 2005, Wiley-VCH, Weinheim. doi : 10.1002/14356007.a23_525
External links

|
Selenium
compounds
|
|
|---|---|
| Se(−II) | |
| Se(0,I) | |
| Se(I) | |
| Se(II) | |
| Se(IV) | |
| Se(VI) | |
| Se(IV,VI) |



