Kornblum–DeLaMare rearrangement
None
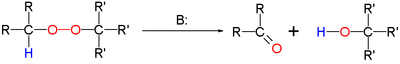
The Kornblum–DeLaMare rearrangement is a rearrangement reaction in organic chemistry in which a primary or secondary organic peroxide is converted to the corresponding ketone and alcohol under acid or base catalysis . The reaction is relevant as a tool in organic synthesis and is a key step in the biosynthesis of prostaglandins . [1]
The base can be a hydroxide such as potassium hydroxide or an amine such as triethylamine .
Reaction mechanism
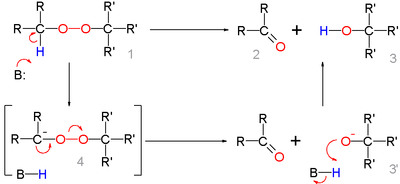
In the reaction mechanism for this organic reaction the base abstracts the acidic α-proton of the peroxide 1 to form the carbanion 4 as a reactive intermediate which rearranges to the ketone 2 with expulsion of the hydroxyl anion 3' . This intermediate gains a proton forming the alcohol 3 .
Deprotonation and rearrangement can also be a concerted reaction without formation of 4 .
An alternative reaction mechanism involving direct nucleophilic displacement on the peroxide link of the amine followed by an elimination reaction is considered unlikely based on the outcome of this model reaction: [2]
The peroxide 1 converts to the hydroxyketone 2 by action of triethylamine but the alternative route through hydroxylamine 3 by nucleophilic displacement with Lithium diisopropylamide and the ammonium salt 4 (by methylation with methyl trifluoromethanesulfonate ) fails.
The reaction, formally a rearrangement, ranks under the elimination reactions as already observed by the original authors. Not only alkoxides but any leaving group capable of carrying a negative charge will do for instance nitrate esters R–C(R)(H)–O–NO 2 .
Related reactions
The corresponding reaction involving an ether is the 1,2-Wittig rearrangement . The reaction course in this rearrangement is different because ether cleavage with carbanion formation is unfavorable. The Pummerer rearrangement in one of its reaction step contains a sulfur variation.
Scope
The original 1951 publication concerned the conversion of potassium t-butyl peroxide and 1-phenylethyl bromide to ultimately acetophenone and t-butanol with piperidine as the base:
The Kornblum–DeLaMare rearrangement can be carried out as an asymmetric reaction with a suitable chiral amine such as sparteine or a cinchona alkaloid: [3]
The first step in this one-pot reaction is 1,4-dioxygenation of 1,3-cycloheptadiene with singlet oxygen and a TPP catalyst.
References
- ↑ The base catalyzed decomposition of a dialkyl peroxide Nathan Kornblum and Harold E. DeLaMare J. Am. Chem. Soc. ; 1951 ; 73(2) pp. 880–81; ( doi : 10.1021/ja01146a542 )
- ↑ The mechanism of the tertiary amine catalysed isomerisation of endoperoxides to hydroxyketones: synthesis and chemistry of the intermediate postulated in the peroxide attack mechanism David R. Kelly, Harjinder Bansal and J. J. Gwynfor Morgan Tetrahedron Letters Volume 43, Issue 51 , 16 December 2002 , Pages 9331–9333 doi : 10.1016/S0040-4039(02)02374-2
- ↑ Enantioselective Synthesis of -Hydroxyenones by Chiral Base-Catalyzed Kornblum DeLaMare Rearrangement Steven T. Staben, Xin Linghu, and F. Dean Toste J. Am. Chem. Soc. ; 2006 ; 128(39) pp. 12658–12659; (Communication) ( doi : 10.1021/ja065464x )