Homologation reaction
Chemical reaction which converts between members of a homologous series
In organic chemistry , a homologation reaction , also known as homologization , is any chemical reaction that converts the reactant into the next member of the homologous series . A homologous series is a group of compounds that differ by a constant unit, generally a methylene ( −CH 2 − ) group. The reactants undergo a homologation when the number of a repeated structural unit in the molecules is increased. The most common homologation reactions increase the number of methylene ( −CH 2 − ) units in saturated chain within the molecule. [1] For example, the reaction of aldehydes or ketones with diazomethane or methoxymethylenetriphenylphosphine to give the next homologue in the series.
Examples of homologation reactions include:
-
Kiliani-Fischer synthesis
, where an
aldose
molecule is elongated through a three-step process consisting of:
- Nucleophillic addition of cyanide to the carbonyl to form a cyanohydrin
- Hydrolysis to form a lactone
- Reduction to form the homologous aldose
- Wittig reaction of an aldehyde with methoxymethylenetriphenylphosphine , which produces a homologous aldehyde.
- Arndt–Eistert reaction is a series of chemical reactions designed to convert a carboxylic acid to a higher carboxylic acid homologue (i.e. contains one additional carbon atom)
- Kowalski ester homologation , an alternative to the Arndt-Eistert synthesis. Has been used to convert β-amino esters from α-amino esters through an ynolate intermediate. [2]
- Seyferth–Gilbert homologation in which an aldehyde is converted to a terminal alkyne and then hydrolyzed back to an aldehyde.
Some reactions increase the chain length by more than one unit. For example, the DeMayo reaction can be considered a two-carbon homologation reaction.
Chain reduction
Likewise the chain length can also be reduced:
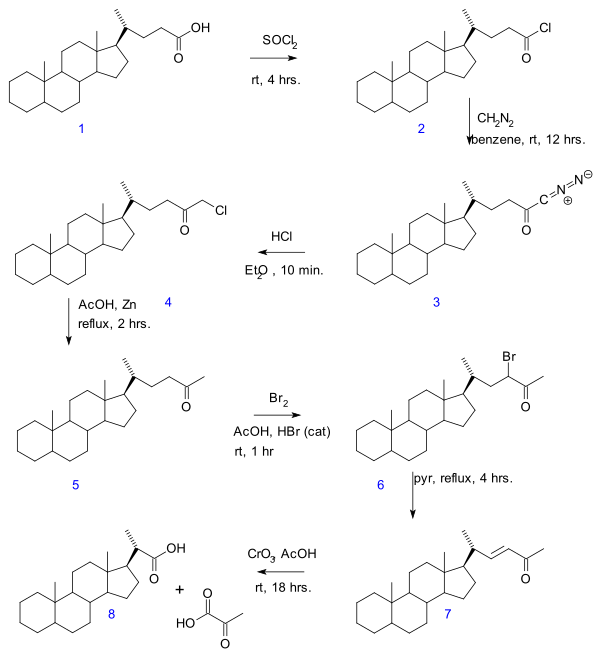
- In the Gallagher–Hollander degradation (1946) pyruvic acid is removed from a linear aliphatic carboxylic acid yielding a new acid with 2 carbon atoms less. [3] The original publication concerns the conversion of bile acid in a series of reactions: acid chloride ( 2 ) formation with thionyl chloride , diazoketone formation ( 3 ) with diazomethane , chloromethyl ketone formation ( 4 ) with hydrochloric acid , organic reduction of chlorine to methylketone ( 5 ), ketone halogenation to 6 , elimination reaction with pyridine to enone 7 and finally oxidation with chromium trioxide to bisnorcholanic acid 8 .
- In the Hooker reaction (1936) an alkyl chain in a certain naphthoquinone (phenomenon first observed in the compound lapachol ) is reduced by one methylene unit as carbon dioxide in each potassium permanganate oxidation . [4] [5]
-

- Mechanistically oxidation causes ring-cleavage at the alkene group, extrusion of carbon dioxide in decarboxylation with subsequent ring-closure.
See also
References
- ↑ Encyclopedia of Inorganic Chemistry doi : 10.1002/0470862106.id396
- ↑ D. Gray, C. Concellon and T. Gallagher (2004). "Kowalski Ester Homologation. Application to the Synthesis of β-Amino Esters". J. Org. Chem. 69 (14): 4849–4851. doi : 10.1021/jo049562h . PMID 15230615 .
- ↑ Vincent P. Hollander and T. F. Gallagher PARTIAL SYNTHESIS OF COMPOUNDS RELATED TO ADRENAL CORTICAL HORMONES. VII. DEGRADATION OF THE SIDE CHAIN OF CHOLANIC ACID J. Biol. Chem. , Mar 1946 ; 162: 549 - 554 Link
- ↑ On the Oxidation of 2-Hydroxy-1,4-naphthoquinone Derivatives with Alkaline Potassium Permanganate Samuel C. Hooker J. Am. Chem. Soc. 1936 ; 58(7); 1174-1179. doi : 10.1021/ja01298a030
- ↑ On the Oxidation of 2-Hydroxy-1,4-naphthoquinone Derivatives with Alkaline Potassium Permanganate. Part II. Compounds with Unsaturated Side Chains Samuel C. Hooker and Al Steyermark J. Am. Chem. Soc. 1936 ; 58(7); pp 1179 - 1181; doi : 10.1021/ja01298a031